2023-06-26 ペンシルベニア州立大学(PennState)
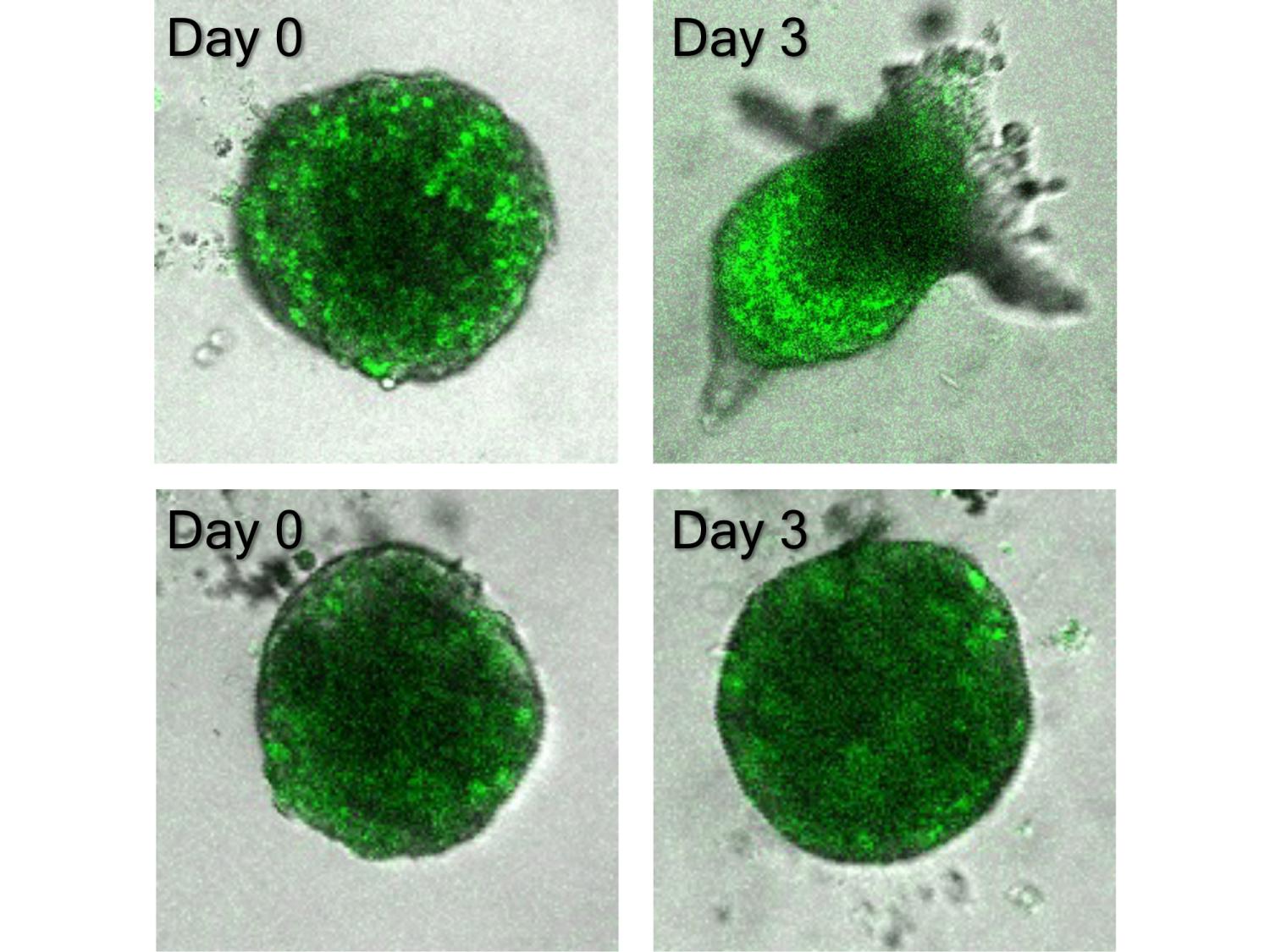 A Penn State-led research team found that a molecule called MALAT1 helps regulate the cells that lead cancer invasion. The top row shows bladder cancer cells with MALAT1 expression, while the bottom row shows bladder cancer cells with reduced MALAT1 expression. In the top row, the cells are beginning their migration away from the tumor, while the bottom row shows no such migration. Credit: Wong Lab/Penn State. All Rights Reserved.
A Penn State-led research team found that a molecule called MALAT1 helps regulate the cells that lead cancer invasion. The top row shows bladder cancer cells with MALAT1 expression, while the bottom row shows bladder cancer cells with reduced MALAT1 expression. In the top row, the cells are beginning their migration away from the tumor, while the bottom row shows no such migration. Credit: Wong Lab/Penn State. All Rights Reserved.
◆彼らはリーダー細胞と呼ばれる特定の細胞を追跡するナノバイオセンサーを使用し、リーダー細胞を特定するための分子マーカーを見つけました。このマーカーは腫瘍の浸潤性を予測するために利用される可能性があります。さらに、研究チームはマラット1という遺伝子がリーダー細胞に関与しており、マラット1の発現ががん細胞の浸潤に重要な役割を果たしていることを発見しました。この研究は、将来的には新しい予後予測ツールや治療法の開発につながる可能性があります。
<関連情報>
- https://www.psu.edu/news/engineering/story/follow-leader-researchers-identify-mechanism-cancer-invasion/
- https://www.pnas.org/doi/10.1073/pnas.2305410120
長鎖ノンコーディングRNA MALAT1が、がんの集団浸潤時にリーダー細胞で動的に制御されることを発見 Long noncoding RNA MALAT1 is dynamically regulated in leader cells during collective cancer invasion
Ninghao Zhu, Mona Ahmed, Yanlin Li, Joseph C. Liao, and Pak Kin Wong
Proceedings of the National Academy of Sciences Published:June 26, 2023
DOI:https://doi.org/10.1073/pnas.2305410120
Significance
The prevalence of collective cancer invasion as a mechanism of cancer metastasis is widely reported, but the regulatory mechanisms at the invading fronts of tumors remain largely unknown. Conflicting studies on the role of long noncoding RNA (lncRNA) MALAT1 (metastasis-associated lung adenocarcinoma transcript 1) in cancer metastasis highlight the complexity and challenges of revealing the dynamic functions of lncRNAs. This study introduces a nanobiosensor that monitors lncRNA dynamics in live single cancer cells and probes the spatiotemporal dynamics of lncRNAs at the invasion fronts of cancer cells and patient-derived spheroids. The findings demonstrate that lncRNA MALAT1 is dynamically regulated during collective cancer invasion, which could lead to the development of prognostic and therapeutic approaches targeting collective cancer invasion.
Abstract
Cancer cells collectively invade using a leader–follower organization, but the regulation of leader cells during this dynamic process is poorly understood. Using a dual double-stranded locked nucleic acid (LNA) nanobiosensor that tracks long noncoding RNA (lncRNA) dynamics in live single cells, we monitored the spatiotemporal distribution of lncRNA during collective cancer invasion. We show that the lncRNA MALAT1 (metastasis-associated lung adenocarcinoma transcript 1) is dynamically regulated in the invading fronts of cancer cells and patient-derived spheroids. MALAT1 transcripts exhibit distinct abundance, diffusivity, and distribution between leader and follower cells. MALAT1 expression increases when a cancer cell becomes a leader and decreases when the collective migration process stops. Transient knockdown of MALAT1 prevents the formation of leader cells and abolishes the invasion of cancer cells. Taken together, our single-cell analysis suggests that MALAT1 is dynamically regulated in leader cells during collective cancer invasion.

