2026-04-16 浙江大学(ZJU)
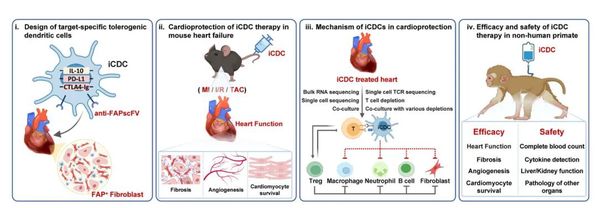
Schematic of iCDC engineering and the experimental workflow. Therapeutic efficacy and mechanisms were evaluated in mouse models of MI, I/R injury and TAC, and translational efficacy and safety were assessed in an NHP MI model.
<関連情報>
- https://www.zju.edu.cn/english/2026/0416/c19573a3152540/page.psp
- https://www.nature.com/articles/s41586-026-10346-5
遺伝子操作された免疫抑制性樹状細胞が心臓リモデリングから保護する Engineered immunosuppressive dendritic cells protect against cardiac remodelling
Xiaoying Li,Jiamin Li,Guohua Li,Lisheng Zhu,Guo Cheng,Huanqiang Li,Hao Lin,Ningqing Jia,Xiaoqian Hong,Ye Liu,Zhiwei Zhong,Yize Chen,Biqing Wang,Jing Zhao,Zhenqi Hua,Lingjun Wang,Qiming Chen,Peijie Zheng,Shuyuan Sheng,Songting Gu,Cheng Ni,Shuchang Ye,Changle Ke,Feimu Zhang,… Xinyang Hu
Nature Published:08 April 2026
DOI:https://doi.org/10.1038/s41586-026-10346-5
Abstract
Heart failure remains a leading cause of morbidity and mortality, yet no approved therapies effectively prevent or reverse pathological cardiac fibrosis and the associated decline in cardiac function1,2,3,4. Chronic inflammation is a central driver of pathological fibrosis after ischaemic or haemodynamic stress, but strategies that locally rebalance injurious and reparative immune responses without systemic immunosuppression are lacking5,6. Dendritic cells (DCs) are key regulators of immune activation and tolerance, providing an opportunity for therapeutic immune reprogramming in cardiac diseases7,8. Here we show that engineered immunosuppressive and fibrosis-targeted DCs (iCDCs) effectively protect against pathological cardiac remodelling. In mouse models of ischaemia–reperfusion injury, myocardial infarction and pressure overload, iCDC therapy reduced inflammatory cardiac fibrosis, improved cardiac perfusion and preserved contractility. Mechanistically, iCDCs conferred sustained cardioprotection directly by suppressing immune and stromal cell activation or indirectly through promoting clonal expansion of regulatory T cells. Importantly, in a non-human primate model of myocardial infarction, iCDC therapy also reduced cardiac fibrosis, improved cardiac perfusion and contractile function without inducing systemic toxicity. These findings establish lesion-targeted immune modulation as a feasible strategy to control cardiac fibrosis and identify engineered dendritic cells as a promising therapeutic platform for treating cardiac remodelling and heart failure.

