2026-03-31 東京大学
転写因子Foxp3とBATFの協調によるエフェクター制御性T細胞分化
<関連情報>
- https://www.u-tokyo.ac.jp/focus/ja/press/z0111_00101.html
- https://www.u-tokyo.ac.jp/content/400283766.pdf
- https://www.cell.com/immunity/abstract/S1074-7613(26)00116-0
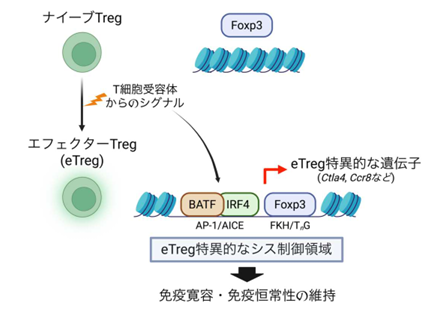
Foxp3とBATFは協調してエフェクターTreg細胞分化のためのシス調節プログラムと遺伝子発現を制御する Foxp3 and BATF cooperatively direct cis-regulatory programs and gene expression for effector Treg cell differentiation
Ryuichi Murakami ∙ Norihito Hayatsu ∙ Takahisa Miyao ∙ … ∙ Haruhiko Koseki ∙ Tomohiro Kurosaki ∙ Shohei Hori
Immunity Published:March 30, 2026
DOI:https://doi.org/10.1016/j.immuni.2026.03.005
Highlights
- Foxp3 and BATF are jointly required for TCR-driven eTreg development
- Foxp3 and BATF synergistically induce eTreg CREs and cis-regulatory programs
- Chromatin accessibility is enhanced at eTreg CREs co-bound by Foxp3 and BATF
- cTreg-specific Foxp3 binding is associated with reduced Tconv CRE accessibility
Summary
Mechanisms by which diverse transcription factors (TFs), particularly the master regulator Foxp3, shape the heterogeneous transcriptional and epigenetic landscapes of regulatory T (Treg) cells remain poorly understood. Here, we show that Foxp3 cooperates with BATF to direct cis-regulatory programs and gene expression essential for the differentiation of immunosuppressive effector Treg (eTreg) cells. Simultaneous single-cell chromatin accessibility and transcriptome profiling, combined with topic modeling, identified cis-regulatory elements and associated programs jointly regulated by these TFs in eTreg cells. Genome-wide mapping of Treg-specific BATF and eTreg-specific Foxp3 binding sites revealed their co-binding at some of these cis-elements, synergistically enhancing accessibility and transcription. Furthermore, we provide evidence that Foxp3 cooperates with or counteracts specific TFs to orchestrate diverse cis-regulatory programs across Treg differentiation states. Thus, Foxp3 serves as a master but context-dependent regulator, cooperating with other TFs, including BATF, to shape the heterogeneous cis-regulatory and transcriptional landscapes critical for functional Treg cell differentiation.