2026-04-10 ジョージア工科大学
<関連情報>
- https://research.gatech.edu/researchers-use-light-make-their-microscopic-muscle-contract-command
- https://coe.gatech.edu/news/2026/04/researchers-use-light-make-their-microscopic-muscle-contract-command?utm_source=twitter&utm_medium=social&utm_campaign=news
- https://www.nature.com/articles/s41467-026-69651-2
光誘起によるCa2+駆動型化学機械的タンパク質ネットワークの集合と再現可能な作動 Light-induced assembly and repeatable actuation in Ca2+-driven chemomechanical protein networks
Xiangting Lei,Carlos Floyd,Laura Casas-Ferrer,Tuhin Chakrabortty,Nithesh Chandrasekharan,Aaron R. Dinner,Scott Coyle,Jerry Honts & Saad Bhamla
Nature Communications Published:21 February 2026
DOI:https://doi.org/10.1038/s41467-026-69651-2
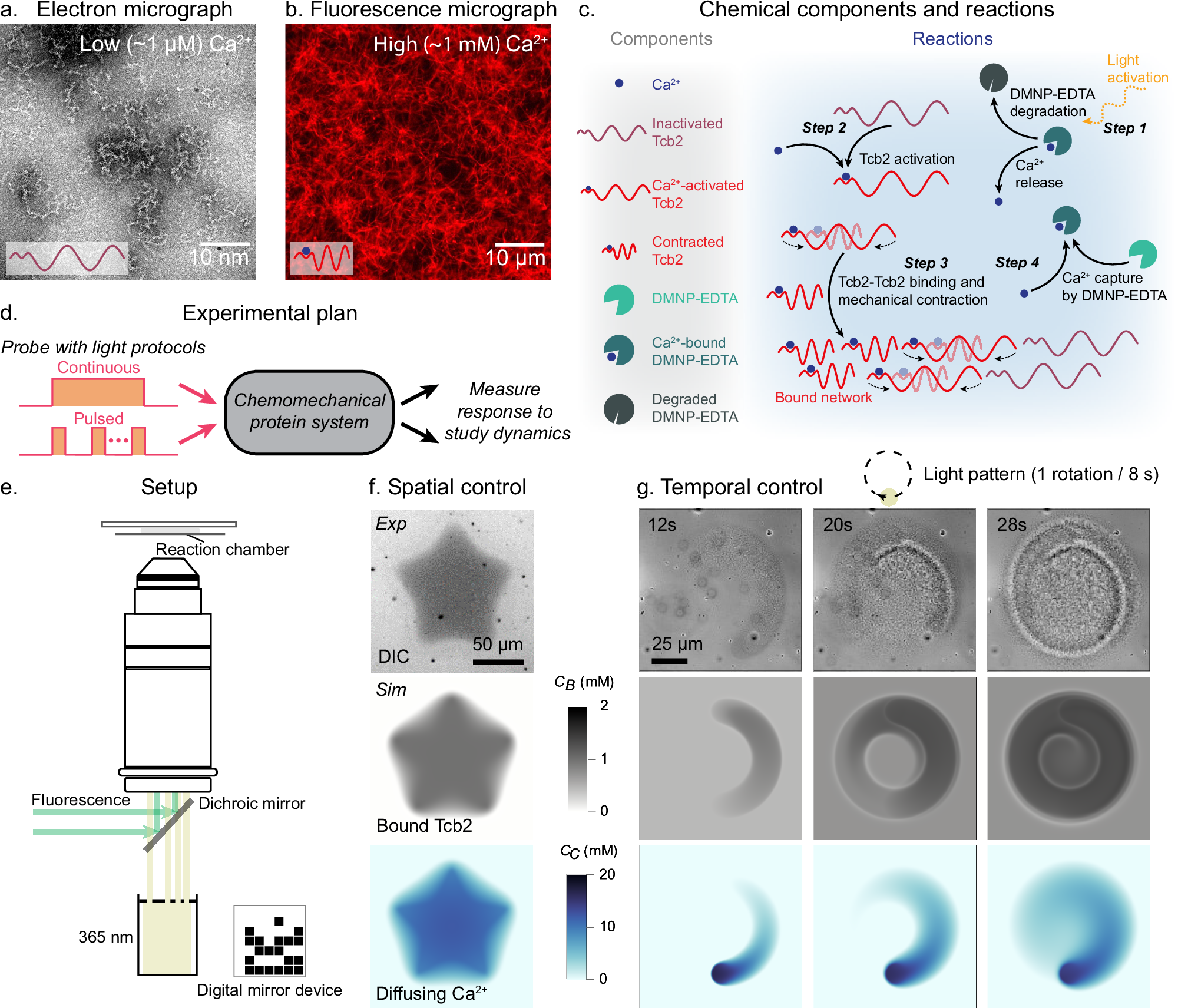
Abstract
Programming rapid, repeatable motions in soft materials has remained a challenge in active matter and biomimetic design. Here, we present a light-controlled chemomechanical network based on Tetrahymena thermophila calcium-binding protein 2 (Tcb2), a Ca2+-sensitive contractile protein. These networks—driven by Ca2+-triggered structural rearrangements—exhibit dynamic self-assembly, spatiotemporal growth, and contraction rates comparable to actomyosin systems. By coupling light-sensitive chelators for optically triggered Ca2+ release, we achieve precise growth and repeatable mechanical contractility of Tcb2 networks, revealing emergent phenomena such as boundary-localized active regions and density gradient-driven reversals in motion. A coupled reaction-diffusion and elastic model explains these dynamics, highlighting the interplay between chemical network assembly and mechanical response. We further demonstrate active transport of particles via network-mediated forces in vitro and implement reinforcement learning to program seconds-scale spatiotemporal actuation in silico. These results establish a platform for designing responsive active materials with rapid chemomechanical dynamics and tunable optical control, with applications in synthetic cells, sub-cellular force generation, and programmable biomaterials.


