~Foxp3は生体内環境に応じてT細胞を再プログラムし、制御性T細胞のエピゲノムと機能を形成する~
2026-05-07 東京大学
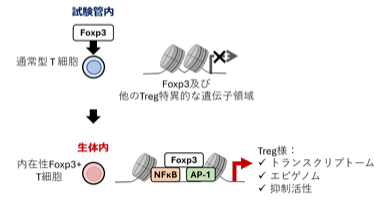
Foxp3 導入によるTregの分子プログラムと機能の形成
<関連情報>
- https://www.u-tokyo.ac.jp/focus/ja/press/z0111_00065.html
- https://www.u-tokyo.ac.jp/content/400287311.pdf
- https://www.science.org/doi/10.1126/sciimmunol.aed2111
Foxp3は、制御性T細胞の分子的なアイデンティティと機能を規定する、状況依存的なエピジェネティックプログラムを促進する Foxp3 drives context-dependent epigenetic programs that define regulatory T cell molecular identity and function
Yuxi Wei, Hinako Ago, Ryuichi Murakami, Shotaro Funatsu, […] , and Shohei Hori
Science Immunology Published:1 May 2026
DOI:https://doi.org/10.1126/sciimmunol.aed2111
Editor’s summary
Regulatory T (Treg) cells are key orchestrators of immunological tolerance and are controlled by the transcription factor Foxp3. However, whether Foxp3 expression alone is sufficient to confer full Treg cell identity remains unclear. Using adoptive transfer of Foxp3-transduced conventional T cells into lymphopenic mice, Wei et al. found that a subset of cells acquired Treg cell–like transcriptional, chromatin, and suppressive features, which required induction of endogenous Foxp3 expression. Acquisition of endogenous Foxp3 occurred in vivo but not in vitro because of elevated AKT-mTOR activity and depended on Foxp3-mediated engagement of STAT5 and NF-κB. These findings support a model in which Foxp3 integrates both intrinsic and environmental cues to confer Treg cell identity and function. —Claire Olingy
Abstract
Regulatory T cells (Treg cells) express the master regulator, Foxp3, and display distinctive epigenetic landscapes ensuring Treg cell–specific gene expression and stable suppressive functions, yet Foxp3’s contribution to this epigenetic identity remains unclear. Leveraging Foxp3-transduced conventional T cells as a gain-of-function probe in mice, we identified a previously unrecognized subset that acquires endogenous Foxp3 expression, Treg cell–like transcriptomic and chromatin features, and suppressive functions exclusively in vivo. These Foxp3-driven features were conserved in Treg cells but impaired in Foxp3-mutant Treg-like cells, demonstrating a Foxp3 requirement. Induction of endogenous Foxp3 expression in vivo required reduced AKT-mTOR signaling and Foxp3-dependent engagement of STAT5 and nuclear factor κB (NF-κB). Temporal chromatin profiling revealed stepwise Foxp3-driven regulatory programs, including a core program shared across Treg cell subsets and effector-specific programs, both associated with NF-κB activity and Foxp3 binding. Thus, Foxp3 integrates cell-intrinsic and environmental contexts to drive epigenetic programs defining Treg cell identities and functions, with implications for Foxp3-based therapies.