2026-02-24 京都大学iPS細胞研究所
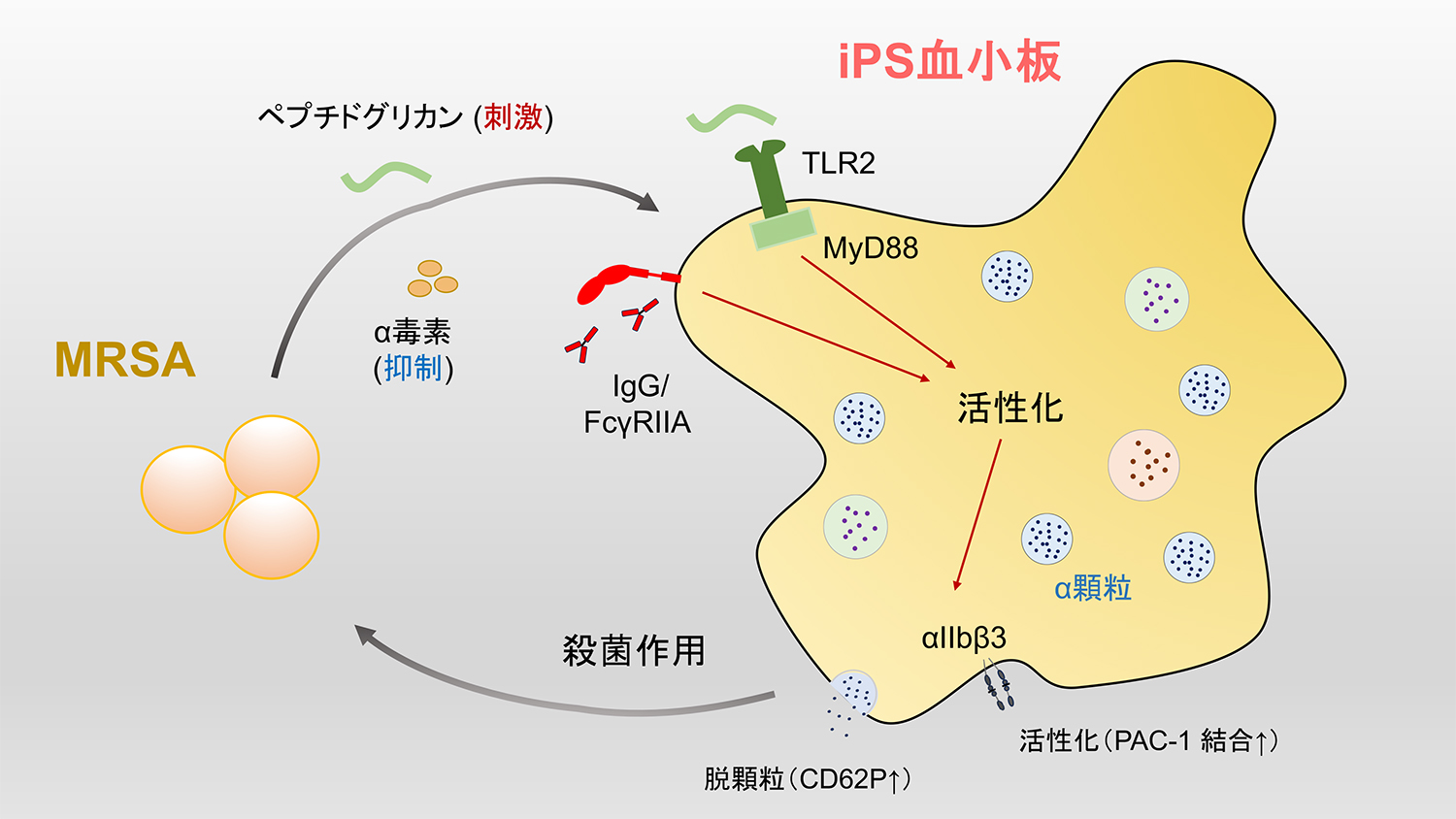
論文の概要図
MRSAのTLR2リガンド成分(PGNなど)は血小板上のTLR2に結合し、アダプタータンパク質MyD88を介したシグナル伝達により血小板を活性化させる(PAC-1結合およびCD62P発現の増加により観測される)。血漿に含まれる免疫グロブリンIgGもFcγR結合を介してこの活性化に寄与し、MRSAに対する殺菌作用を発揮する。他方、MRSAのα毒素は殺菌作用を抑制する働きがある。
<関連情報>
- https://www.cira.kyoto-u.ac.jp/j/pressrelease/news/260224-100000.html
- https://www.rpthjournal.org/article/S2475-0379(26)00032-4/fulltext
人工多能性幹細胞由来血小板は、Toll様受容体2-MyD88シグナル伝達と免疫グロブリンG/FcγRIIAの関与を介して多剤耐性黄色ブドウ球菌を殺菌する Induced pluripotent stem cell-derived platelets kill multidrug-resistant Staphylococcus aureus via Toll-like receptor 2-MyD88 signaling and immunoglobulin G/FcγRIIA engagement
Qirui Lin ∙ Kimiko Nonomura ∙ Ieva Stirblyte ∙ … ∙ Victor Nizet ∙ Koji Eto ∙ Naoshi Sugimoto
Research and Practice in Thrombosis and Haemostasis Published:February 2, 2026
DOI:https://doi.org/10.1016/j.rpth.2026.103374
Essentials
- The MRSA-killing capacity of iPSC-PLTs remains unaddressed.
- We performed in vitro MRSA-killing assays, applying inhibitors, ΔHla MRSA, and MyD88-deficient iPSC-PLTs.
- iPSC-PLTs killed MRSA involving activation, IgG-FcγR and TLR-MyD88 axis, but suppressed by Hla.
- iPSC-PLTs are applicable for MRSA infections and gene ediatable for investigation purposes.
Abstract
Background
Severe infection caused by multidrug-resistant bacteria, such as multidrug-resistant Staphylococcus aureus (MRSA), represents a pressing clinical challenge. While platelets are known to possess antibacterial activity against MRSA, the underlying mechanisms remain incompletely understood. In addition, the MRSA-killing capacity of induced pluripotent stem cell-derived platelets (iPSC-PLTs), which we succeeded in producing ex vivo, has not been previously characterized.
Objectives
We aimed to verify whether iPSC-PLTs are capable of killing MRSA and further elucidate the mechanisms involved in this process.
Methods
We performed in vitro colony assays to assess MRSA killing by iPSC-PLTs. To gain mechanistic insights, we applied antiplatelet agents, an FcγRIIA-blocking antibody, α-toxin–deficient MRSA, and MyD88-deficient iPSC-PLTs, which we created by gene editing.
Results
All 3 iPSC-PLT clones demonstrated MRSA-killing capacity. Although only minimal activation of iPSC-PLTs was observed, antiplatelet agents inhibited this killing. Notably, plasma components enhanced the bactericidal activity of iPSC-PLTs, in part via immunoglobulin G, as evidenced by inhibition with an FcγRIIA-blocking antibody. Compared with wild-type MRSA, α-toxin–deficient strains were more susceptible to iPSC-PLT–mediated killing, suggesting that α-toxin acts as a suppressor of this platelet function. Furthermore, MyD88-deficient iPSC-PLTs exhibited impaired MRSA-killing capacity, indicating the indispensable role of Toll-like receptor 2-mediated signaling in this response.
Conclusion
Collectively, our findings highlight the direct antimicrobial potential of iPSC-PLTs and provide mechanistic insights, particularly into the contribution of the Toll-like receptor–MyD88 axis. This study provided a basis for applying iPSC-PLTs as a novel therapeutic modality for combating MRSA infections and a genetically modifiable platform for investigating unknown platelet function within the context of antimicrobial immunity.