2026-02-26 名古屋大学
<関連情報>
- https://www.nagoya-u.ac.jp/researchinfo/result/2026/02/post-952.html
- https://www.nagoya-u.ac.jp/researchinfo/result/upload_images/20260226_med_jp.pdf
- https://www.jci.org/articles/view/198708
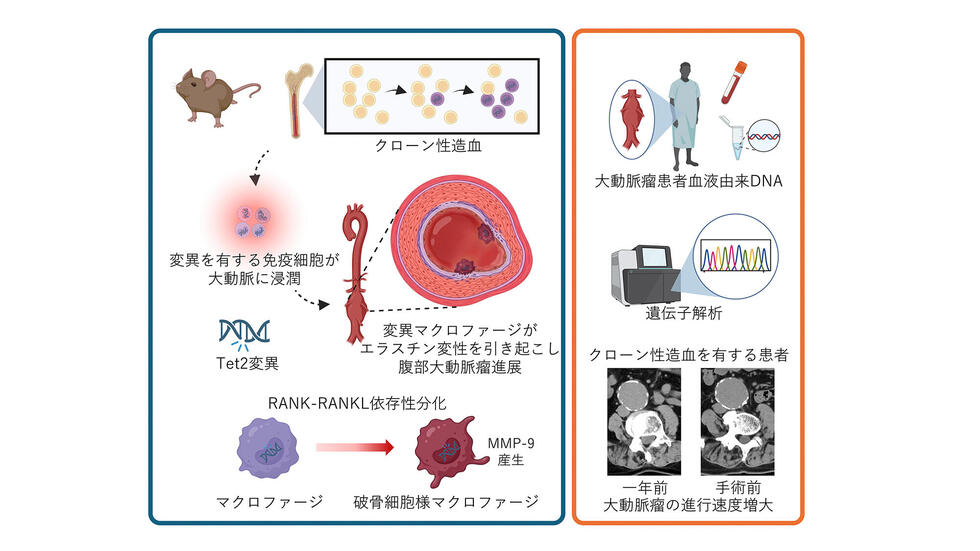
Tet2誘導性クローン造血はマクロファージから破骨細胞様分化を介して大動脈瘤を引き起こす Tet2-driven clonal hematopoiesis drives aortic aneurysm via macrophage-to-osteoclast-like differentiation
Jun Yonekawa, Yoshimitsu Yura, Junmiao Luo, Katsuhiro Kato, Shuta Ikeda, Yohei Kawai, Tomoki Hattori, Ryotaro Okamoto, Mari Kizuki, Emiri Miura-Yura, Keita Horitani, Kyung-Duk Min, Takuo Emoto, Hiroshi Banno, Mikito Takefuji, Kenneth Walsh, and Toyoaki Murohara
Journal of Clinical Investigation Published: February 25, 2026
DOI:https://doi.org/10.1172/JCI198708
Abstract
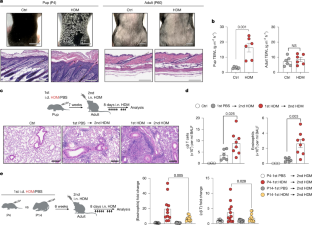
Aortic aneurysms are age-linked aortic dilations that progress silently and carry high rupture mortality. Immune cells are recognized drivers of aneurysm pathogenesis. Clonal hematopoiesis is an age-related expansion of somatically mutated hematopoietic stem cells that reshapes immune function and contributes to diverse age-associated diseases. However, its contribution to aneurysm pathogenesis remains unclear. In this study, targeted ultradeep sequencing of patient specimens revealed a high prevalence of clonal hematopoiesis-associated mutations that correlated with faster aneurysm expansion. Thus, we modeled clonal hematopoiesis by competitively transplanting Tet2-deficient bone marrow into ApoE-knockout mice and induced aneurysms with angiotensin II. Tet2-clonal hematopoiesis mice developed significantly greater aortic dilation than controls. Interestingly, Tet2-deficient macrophages adopted an ACP5-positive, osteoclast-like state and produced more MMP9. Both genetic and pharmacological inhibition of osteoclast-like differentiation suppressed the Tet2-mediated aneurysmal growth in vivo. Thus, Tet2-driven clonal hematopoiesis accelerates aortic aneurysm progression through MMP9-producing osteoclast-like macrophages and therefore represents a tractable therapeutic axis.