- 尿路上皮がんに対する既承認薬エンホルツマブ ベドチンが標的とする腫瘍抗原が小腸腺がんでも高発現していることを発見した研究成果を臨床に展開
- 小腸腺癌における新規治療標的の探索:クローディン18.2、ネクチン4、HER3発現解析からの知見 Exploring novel therapeutic targets in small bowel adenocarcinoma: insights from claudin 18.2, nectin-4, and HER3 expression analysis
- 転移性または切除不能な小腸腺癌の日本人患者を対象とした5-フルオロウラシル/L-ロイコボリン/オキサリプラチン(mFOLFOX6)の第II相試験 A phase II study of 5-fluorouracil/L-leucovorin/oxaliplatin (mFOLFOX6) in Japanese patients with metastatic or unresectable small bowel adenocarcinoma
尿路上皮がんに対する既承認薬エンホルツマブ ベドチンが標的とする腫瘍抗原が小腸腺がんでも高発現していることを発見した研究成果を臨床に展開
2026-03-02 国立がん研究センター,大阪国際がんセンター,九州大学病院
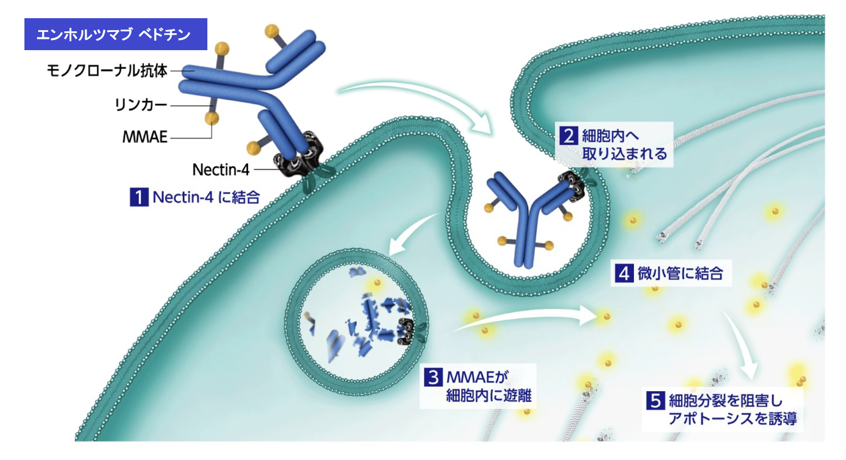
出典:アステラス製薬株式会社社内報告書一部改変:癌細胞(ヒト由来)・薬理作用(ヒトnectin-4への結合親和性)
図1 エンホルツマブ ベドチンは、がん細胞の細胞膜上に発現するNectin-4に結合し、細胞内に取り込まれた後にリンカーが切断され、MMAE(Monomethyl auristatin E;抗がん剤)が細胞内に遊離することでがんの増殖を抑制します。
<関連情報>
- https://www.ncc.go.jp/jp/information/pr_release/2026/0302/index.html
- https://www.esmoopen.com/article/S2059-7029(24)01869-6/fulltext
- https://link.springer.com/article/10.1007/s10147-017-1138-6
小腸腺癌における新規治療標的の探索:クローディン18.2、ネクチン4、HER3発現解析からの知見 Exploring novel therapeutic targets in small bowel adenocarcinoma: insights from claudin 18.2, nectin-4, and HER3 expression analysis
H. Fujii, ∙ H. Shoji ∙ H. Hirano ∙ … ∙ N. Okita ∙ A. Takashima ∙ K. Kato
ESMO Open Published:January 3, 2025
DOI:https://doi.org/10.1016/j.esmoop.2024.104098
Highlights
- Claudin 18.2, nectin-4, and HER3 were expressed in 35%, 82%, and 88% of SBA, respectively.
- The median PD-L1 CPS in SBA was 3 (range 0-60), with CPS <1, 1-5, and ≥5 in 33%, 32%, and 35% of cases, respectively.
- PD-L1 CPS tended to be higher in nectin-4-positive SBA samples (median CPS 2.5 versus 0, P = 0.077).
- Nectin-4-positive patients had significantly shorter OS than negative ones (12.6 versus 43.2 months, P = 0.006, HR 5.12).
- Claudin 18.2, nectin-4, and HER3 are potential therapeutic targets for many SBA patients, warranting further research.
Abstract
Background
Small bowel adenocarcinoma (SBA) is a rare malignancy with few established chemotherapy options and a dismal prognosis. We investigated the expression of claudin 18.2, nectin-4, human epidermal growth factor receptor 3 (HER3), and programmed death-ligand 1 (PD-L1) in SBA to identify potential antibody drug targets and analyzed associated clinicopathological features and prognosis.
Materials and methods
We retrospectively reviewed patients diagnosed with SBA who underwent adjuvant or palliative chemotherapy at our hospital between July 2010 and July 2023. Pathological samples were immunohistochemically stained for claudin 18.2, nectin-4, HER3, and PD-L1. Overall survival (OS) was assessed in patients receiving palliative chemotherapy to examine its association with the expression of each protein, excluding those with microsatellite instability-high who were treated with immunotherapy.
Results
Pathological samples and clinical data were available for 51 patients. The primary lesion was in the duodenum in 49% of these patients and in the jejunum or ileum in 51%. Positive rates for claudin 18.2, nectin-4, and HER3 were 35%, 82%, and 88%, respectively. All cases expressed at least one of the proteins, and 25% expressed all three proteins. The PD-L1 combined positive score (CPS) was <1, 1-5, and ≥5 in 33%, 32%, and 35%, respectively; nectin-4-positive samples showed higher CPS. Neither claudin 18.2 nor HER3 positivity was associated with OS. However, nectin-4 positivity was associated with significantly shorter OS [12.6 versus 43.2 months, hazard ratio (HR) 5.12, P = 0.006]. Similarly, PD-L1 CPS ≥5 was associated with shorter OS relative to CPS <5 (9.7 versus 18.0 months, HR 2.60, P = 0.028). Multivariate analysis identified nectin-4 positivity (HR 4.55, P = 0.020) as an independent adverse prognostic factor for OS.
Conclusions
Claudin 18.2, nectin-4, and HER3 are potential therapeutic targets in SBA, and nectin-4 positivity is independently associated with an unfavorable prognosis. These proteins may represent new therapeutic targets for SBA.
転移性または切除不能な小腸腺癌の日本人患者を対象とした5-フルオロウラシル/L-ロイコボリン/オキサリプラチン(mFOLFOX6)の第II相試験 A phase II study of 5-fluorouracil/L-leucovorin/oxaliplatin (mFOLFOX6) in Japanese patients with metastatic or unresectable small bowel adenocarcinoma
Takahiro Horimatsu,Norisuke Nakayama,Toshikazu Moriwaki,Yoshinori Hirashima,Mikio Fujita,Masako Asayama,Ichiro Moriyama,Koji Nakashima,Eishi Baba,Hiroshi Kitamura,Takao Tamura,Ayumu Hosokawa,Kenichi Yoshimura & Manabu Muto
International Journal of Clinical Oncology Published:23 May 2017
DOI:https://doi.org/10.1007/s10147-017-1138-6
Abstract
Background
Several studies have suggested that chemotherapy prolonged survival in patients with metastatic or recurrent small bowel adenocarcinoma (SBA); however, there is still no standard chemotherapy regimen. Here, we evaluated the efficacy and safety of a 5-fluorouracil (5-FU)/L-leucovorin (l-LV)/oxaliplatin (mFOLFOX6) protocol as a first-line therapy for patients with SBA.
Patients and methods
This was a multicenter, single-arm, open-label phase II study. Eligibility criteria included histologically confirmed adenocarcinoma, age 20–80 years, and an Eastern Cooperative Oncology Group performance status (PS) of 0–2. The primary endpoint was 1-year progression-free survival (PFS). The secondary endpoints included overall response rate (ORR), overall survival (OS), overall PFS, and safety.
Results
Between April 2010 and November 2012, 24 patients were enrolled from 12 institutions. The median age of the patients was 63 years (range 31–79) and there was a male/female ratio of 18/6. The number of PS 0/1 patients was 17/7 and locally advanced/metastatic disease was seen in 2/22 patients, respectively. The primary tumor site was the duodenum in 14 patients (58%) and jejunum in 10 patients (42%). The median follow-up time was 14.7 months (3.7–40.3). The 1-year PFS was 23.3%. The ORR was 9/20 (45%). The median PFS and OS times were 5.9 months (95% confidence interval [CI] 3.0–10.2) and 17.3 months (95% CI 11.7–19.0), respectively. Major grade 3/4 toxicities were neutropenia (38%), anemia/peripheral neuropathy (25%), and stenosis (17%). There were no treatment-related deaths.
Conclusions
Although the primary endpoint was not met, mFOLFOX6 showed effective and good tolerance as a first-line treatment for SBA.