2026-03-23 米国国立再生可能エネルギー研究所(NREL)
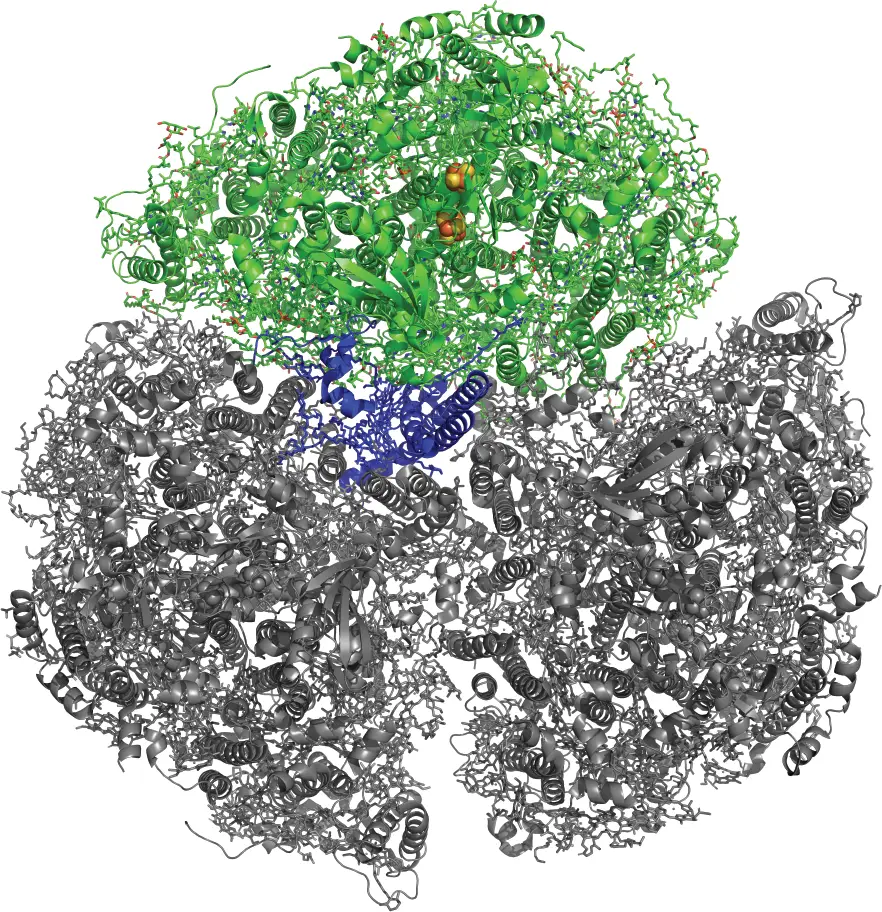
Photosystem I (PSI), a large protein-pigment complex, resides within cyanobacteria to absorb light and convert it into chemical energy. One monomeric unit of the trimeric structure is shown in color (upper third). The blue portion is the PsaL subunit that is the focus of this research. Rendering by Cara Lubner, National Laboratory of the Rockies
<関連情報>
- https://www.nlr.gov/news/detail/program/2026/researchers-describe-protein-structure-microbes-use-to-control-light-conversion
- https://www.jbc.org/article/S0021-9258(25)02083-6/fulltext
電子の流れ経路と光子束の変化に応じたシアノバクテリアの光化学系Iタンパク質環境とスペクトル容量の調節 Modulation of cyanobacterial Photosystem I protein environment and spectral capacity in response to changes in electron flow pathways and photon flux
Sharon L. Smolinski ∙ Monika Tokmina-Lukaszewska ∙ Junia M. Holland ∙ … ∙ Brian Bothner ∙ Paul W. King ∙ Carolyn E. Lubner
Journal of Biological Chemistry Published online May 14, 2025
DOI:https://doi.org/10.1016/j.jbc.2025.110233
Abstract
Cyanobacterial photosystem I (PSI) can undergo modifications that adjust photosynthetic electron transport in response to fluctuations in environmental and cellular conditions. We recently reported that PSI isolated from Synechocystis sp. PCC 6803 (S. 6803) strains lacking a peripheral oxygen reduction reaction (ORR1) pathway demonstrated altered P700 photooxidation capacity, changes in spectral properties, and a higher proportion of monomers. These changes in PSI were augmented when cells were grown under higher photon flux, which creates a greater energy imbalance at PSI. We have shown that the modified PSI is functional in photochemical charge separation and ferredoxin reduction reactions. Thus, we hypothesized that monomerization of PSI was caused by changes in the environment of PsaL, which is known to be essential for stabilizing trimers. To test our hypothesis, we isolated PSI monomers and trimers from ORR1 and wild-type (WT) strains. The electron paramagnetic resonance (EPR) spectra of reduced PSI demonstrated the presence of intact FA and FB [4Fe-4S] clusters, consistent with measurements of functional charge separation and electron transport. Limited proteolysis followed by mass spectrometric analysis showed altered accessibility of PsaL in the ORRI PSI monomers compared to WT monomers, and included regions associated with chlorophyll and carotenoid binding, and in functional interactions with adjacent subunits. In addition, ORR1 PSI monomers had spectral changes compared to WT PSI due to differences in carotenoid compositions. Collectively, these findings reveal new insights into how microbes adjust PSI structure and photochemistry to mitigate photodamage in response to changes in electron utilization by downstream chemical reactions.

