20205-04-21 東京大学
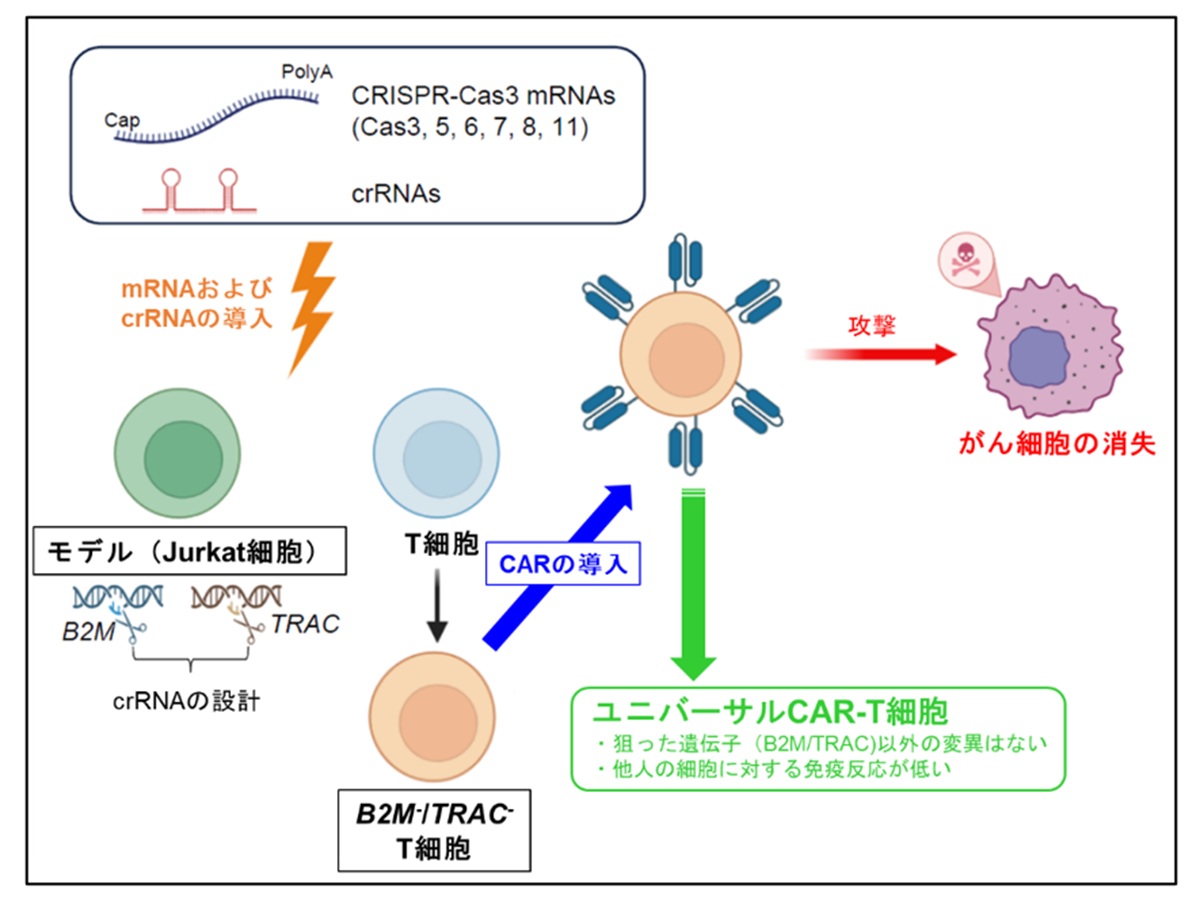
CRISPR-Cas3によるユニバーサルCAR-T細胞作製の概要
<関連情報>
- https://www.ims.u-tokyo.ac.jp/imsut/jp/about/press/page_00390.html
- https://academic.oup.com/narcancer/article/8/2/zcag009/8657738?login=false
ヒトT細胞におけるCRISPR-Cas3を用いた効率的な遺伝子破壊 Efficient gene disruption with CRISPR–Cas3 in human T cells
Tomoaki Fujii,Yukimi Sakoda,Kazuto Yoshimi,Kohei Takeshita,Shoji Watanabe,Ryuya Iida,Tsubasa Obo,Kazumasa Yokoyama,Koji Tamada,Tomoji Mashimo
NAR Cancer Published:21 April 2026
DOI:https://doi.org/10.1093/narcan/zcag009
Abstract
The CRISPR–Cas9 system has been widely adopted as a genome editing tool due to its high efficiency and versatility, contributing to the development of various therapeutic strategies. However, its clinical application remains limited by safety concerns, including off-target effects and large-scale chromosomal rearrangements such as translocations and inversions. Recently, the CRISPR–Cas3 system, a Class 1 CRISPR effector complex with unidirectional DNA degradation activity, has gained attention as a potential alternative, offering reduced off-target activity. In this study, we applied the CRISPR–Cas3 system to human T cells and successfully disrupted two clinically relevant genes, T cell receptor alpha constant (TRAC) and beta-2 microglobulin (B2M). These gene deletions were associated with a reduction in both graft-versus-host disease risk and host immune rejection. Importantly, no off-target mutations were detected in CRISPR–Cas3-edited cells, in contrast to the off-target effects observed with CRISPR–Cas9. Furthermore, CAR-T cells generated by deleting TRAC or B2M using CRISPR–Cas3 maintained their antigen-specific cytotoxicity against tumor cells, while exhibiting reduced alloreactivity. These results suggest that CRISPR–Cas3 provides a safer and promising platform for genome editing in T cell engineering, with potential applications in the development of next-generation allogeneic T cell therapies.