2026-04-28 韓国基礎科学研究院(IBS)
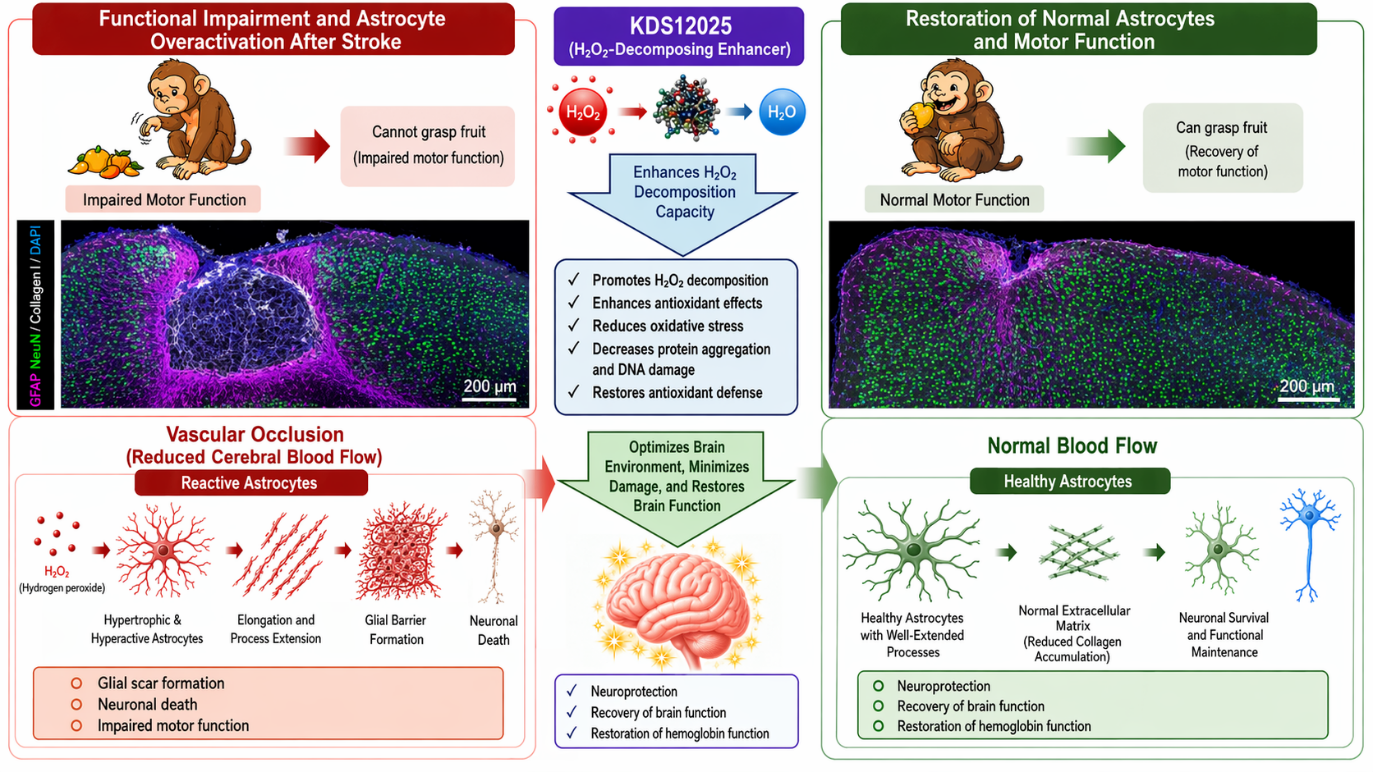
Figure 1. Schematic illustration of astrocyte-derived Type I collagen production and neuronal cell death in ischemic stroke
Following ischemic injury, excessive hydrogen peroxide (H₂O₂) is generated, leading to astrocyte activation. Activated astrocytes produce collagen proteins, which contribute to the formation of a glial barrier and induce neuronal cell death. Importantly, inhibition of collagen production or early elimination of excessive hydrogen peroxide prevents the progression of neuronal damage and restores motor function, as demonstrated in non-human primate models.
<関連情報>
- https://www.ibs.re.kr/cop/bbs/BBSMSTR_000000000738/selectBoardArticle.do?nttId=26653&pageIndex=1&searchCnd=&searchWrd=
- https://www.cell.com/cell-metabolism/fulltext/S1550-4131(26)00139-7
酸化ストレスによって誘発されるアストロサイトのコラーゲン生合成は、虚血性脳卒中におけるグリアバリア形成と神経細胞死を促進する Oxidative stress-induced astrocytic collagen biosynthesis drives glial barrier formation and neuronal death in ischemic stroke
Jae-Hun Lee ∙ In-Young Hwang ∙ Hyun Jun Jang ∙ … ∙ Boyoung Lee ∙ Seungjun Ryu ∙ C. Justin Lee
Cell Metabolism Published:April 27, 2026
DOI:https://doi.org/10.1016/j.cmet.2026.04.001
Highlights
- H2O2 rewires astrocytic metabolism to induce COL1 biosynthesis and barrier formation
- H2O2 regulates miR-29 and FUT8 to coordinate glycosylation-dependent COL1 production
- During ischemic stroke, the glial barrier confines damaged neurons to be exterminated
- Targeting the astrocytic H2O2-COL1 axis prevents neuronal death and aids functional recovery
Summary
Astrocytes regulate brain metabolism and homeostasis, but how oxidative stress reshapes astrocytic metabolism to drive neuronal damage remains unclear. Here, we demonstrate that oxidative stress turns on astrocytic type I collagen (COL1) production via a redox-glycosylation coupling mechanism. Hydrogen peroxide (H2O2) suppresses miR-29 and enhances fucosyltransferase 8 (FUT8)-mediated core fucosylation, integrating post-transcriptional and glycosylation-dependent regulation of COL1. Astrocyte-derived COL1 activates integrin signaling and promotes neuronal death. In a photothrombotic stroke model, an H2O2 surge induces astrogliosis, glycosylation remodeling, and COL1 expression, leading to glial barrier formation, neuronal loss, and neurological deficits. These pathological cascades are mitigated by astrocyte-specific silencing of COL1 or FUT8 or by KDS12025, a peroxidase enhancer that reduces H2O2 burden. Notably, KDS12025 exerts potent neuroprotection in a non-human primate stroke model. Together, our findings identify an unprecedented astrocytic metabolic pathway linking oxidative stress to glycosylation-driven COL1 production, highlighting the H2O2 surge, astrocytic COL1, and FUT8 as promising therapeutic targets for recovery after stroke.