2026-05-12 合肥物質科学研究院(HFIPS)
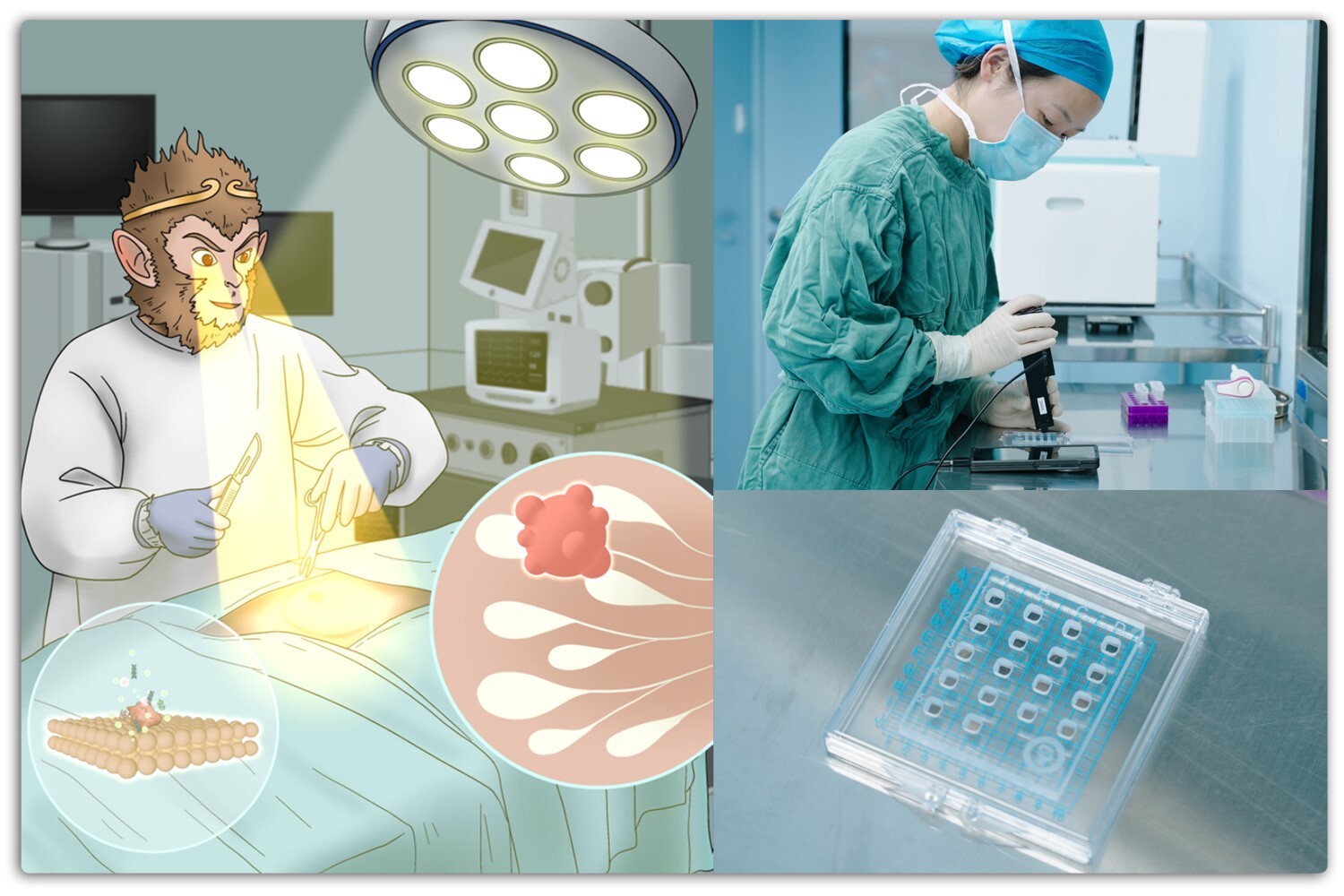
Schematic illustration of the BioGate SERS Chip for rapid intraoperative detection of breast cancer surgical margins. (Image by HUANG Guangyao)
<関連情報>
- https://english.hf.cas.cn/nr/bth/202605/t20260512_1159227.html
- https://link.springer.com/article/10.1186/s12951-026-04468-1
BioGate SERSチップは、乳がんの境界部を代謝物指向型のナノバイオテクノロジーによって高精度に検出することを可能にする BioGate SERS Chip enables robust metabolite-oriented nanobiotechnological detection of breast cancer margins
Guangyao Huang,Sensen Hao,Yue Fang,Jing Pei,Ying Wang,Jingyuan Shao,Lanxin Bao,Zhenxing Li,Wen Cao,Mancun Zhao,Liujie Hou & Hongzhi Wang
Journal of Nanobiotechnology Published:26 April 2026
DOI:https://doi.org/10.1186/s12951-026-04468-1 Unedited version
Abstract
Accurate identification of malignant surgical margins remains a major challenge in breast cancer surgery due to the lack of rapid and molecularly specific diagnostic tools capable of operating in complex biological environments. Surface-enhanced Raman spectroscopy (SERS) holds promise for molecular detection; however, its application in tissues is limited by nonspecific macromolecular adsorption, signal instability, and biological screening effects. Here, we present the BioGate SERS Chip, a nanobiotechnological platform designed to enable robust, metabolite-oriented SERS detection by regulating molecular access to plasmonic hotspots. The chip features a hierarchical gold nanoparticle architecture that forms bioselective nanochannels, allowing low–molecular-weight metabolites to access electromagnetic hotspot regions while excluding macromolecules responsible for biofouling and signal suppression. This bioselective hotspot-gating strategy couples plasmonic field confinement with size-selective molecular transport, enabling reliable metabolite sensing directly in complex tissue environments. The BioGate platform exhibited a detection limit of 10⁻¹³ M for molecular probes and enabled reliable identification of malignant signatures in mixed cell populations containing as little as 1% tumor cells. Across spontaneous tumor models, xenograft surgeries, and intraoperative specimens from 50 breast cancer patients representing three major molecular subtypes (HER2+, TNBC, and HR+), the platform rapidly distinguished malignant from benign tissues within minutes using subtype-independent metabolic fingerprints. Integrated metabolomic and transcriptomic analyses further revealed that the characteristic Raman peaks originate from amino acid–related metabolic pathways, providing biochemical interpretability at the nano–bio interface. Together, these results establish the BioGate SERS Chip as a practical nanobiotechnology strategy for rapid metabolite-based intraoperative margin detection and demonstrate how bioengineered plasmonic architectures can enable robust SERS sensing in complex biological tissues.


