2026-02-25 広島大学
<関連情報>
- https://www.hiroshima-u.ac.jp/news/95980
- https://academic.oup.com/jbmr/advance-article-abstract/doi/10.1093/jbmr/zjaf201/8405722
マウスRunx2 のrunt相同ドメインにおける病原性変異が鎖骨頭蓋骨異形成症の骨格および歯の表現型に 及ぼす機能的影響 Functional impact of pathogenic mutations in the runt homology domain of mouse Runx2 on skeletal and dental phenotypes in cleidocranial dysplasia
Saki Ogawa,Shinnosuke Higuchi,Yuki Yoshimoto,Mari Hoshino,Shigenori Miura,Atsuko Hamada,Hitomi Watanabe,Tetsushi Sakuma,Kadi Hu,Shun Ogata,…
Journal of Bone and Mineral Research Published:27 December 2025
DOI:https://doi.org/10.1093/jbmr/zjaf201
Abstract
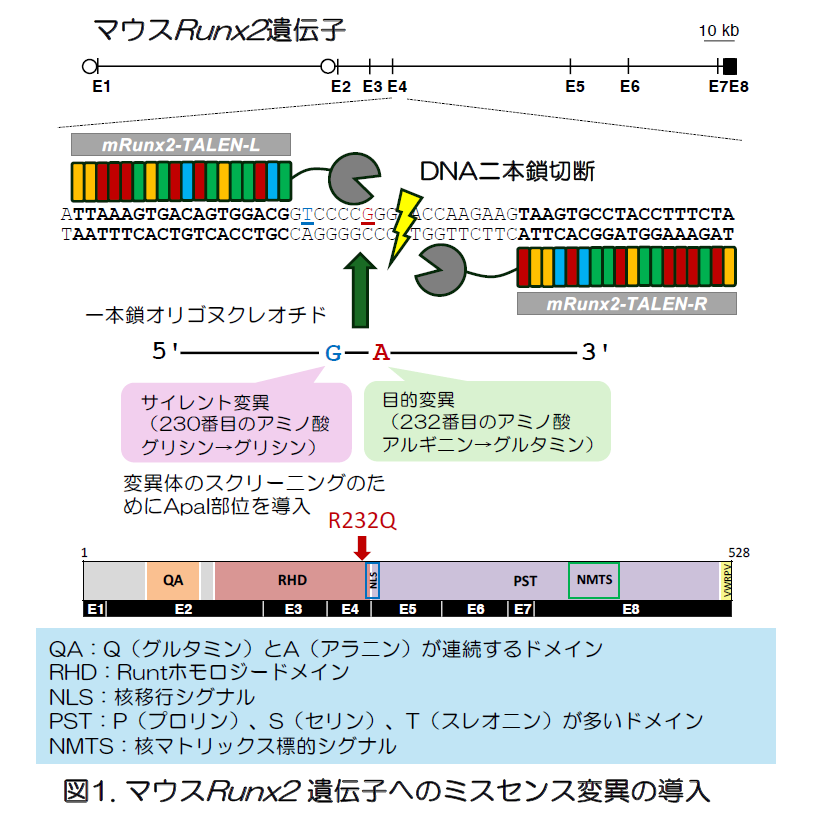
Runt-related transcription factor 2 (RUNX2) is essential for skeletogenesis, and mutations in its gene cause cleidocranial dysplasia (CCD), an autosomal dominant skeletal disorder. The evolutionarily conserved 128-amino acid Runt homology domain (RHD) of human RUNX2 is essential for DNA binding and heterodimerization, and serves as a mutation hotspot associated with severe CCD phenotypes. To elucidate the functional impact of pathogenic RHD mutations in vivo, we generated 2 novel mouse lines: one carrying a missense mutation, c.695G>A, p.Arg232Gln (p.R232Q), corresponding to the human RUNX2 c.674G>A, p.Arg225Gln (p.R225Q), and the other harboring a frameshift mutation, c.697_698delGA, p.Glu233Thrfs*9 (p.E233Tfs*9), causing a premature stop codon. Homozygous Runx2R232Q/R232Q and Runx2E233Tfs*9/E233Tfs*9 mice lacked membranous ossification, whereas heterozygous Runx2R232Q/+ and Runx2E233Tfs*9/+ mice displayed typical CCD-like skeletal features, including an open anterior fontanelle and clavicle hypoplasia. Unexpectedly, heterozygotes carrying pathogenic RHD mutations developed small root-like protrusions, mostly 1 but rarely 2, at the pulp chamber floor of 3-rooted maxillary first molars during furcation, revealing a previously unrecognized dental phenotype. Dual luciferase assays showed that p.R232Q almost completely lost transactivation of the osteocalcin enhancer/promoter. Immunostaining showed that wild-type Runx2 was robustly expressed in osteoblasts and hypertrophic chondrocytes during bone formation, while the p.R232Q mutant Runx2 in Runx2R232Q/R232Q mice exhibited reduced expression in hypertrophic chondrocytes and partially impaired nuclear localization. These abnormalities led to defective osteoblast differentiation and chondrocyte maturation. Thus, our mutant mouse model provides a valuable in vivo platform to study CCD pathogenesis, mechanisms of tooth root furcation, and therapeutic interventions targeting dysfunctional RHD.