2026-02-27 東京科学大学
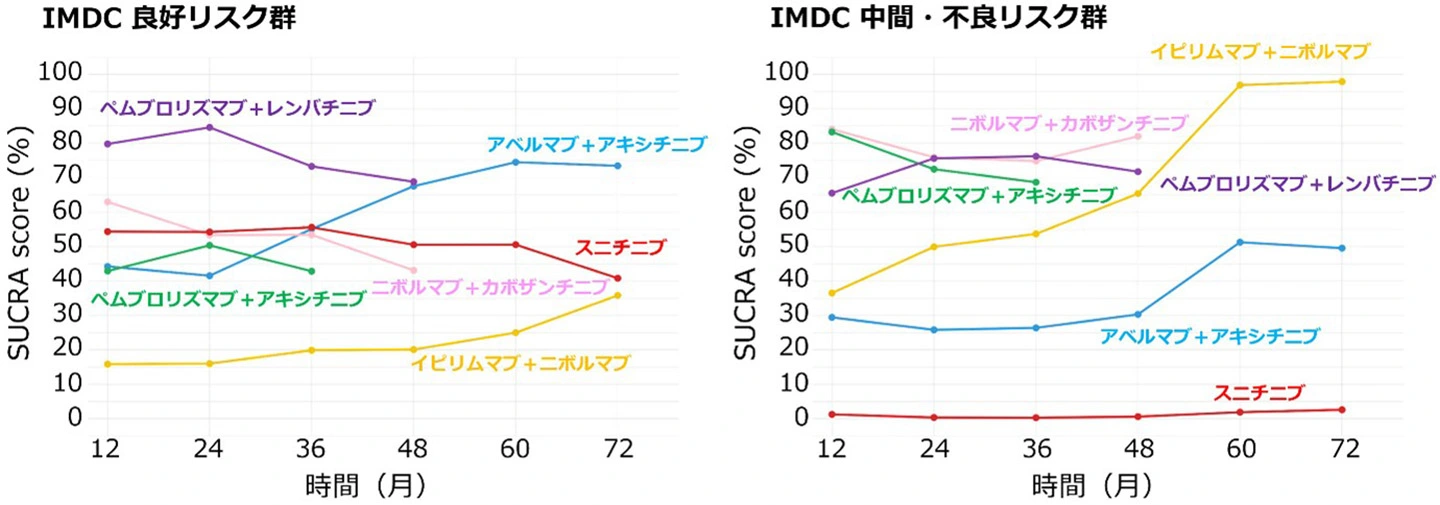
図1. IMDCリスク分類ごとの治療効果の経時的変化
<関連情報>
- https://www.isct.ac.jp/ja/news/0n2nqbxfsmac#top
- https://link.springer.com/article/10.1007/s11523-025-01194-w
転移性明細胞腎細胞癌に対する第一選択治療の時間依存的比較効果:制限付き平均生存期間時間ベースネットワークメタアナリシス Time-Dependent Comparative Effectiveness of First-Line Treatment for Metastatic Clear Cell Renal Cell Carcinoma: A Restricted Mean Survival Time-Based Network Meta-analysis
Hiroshi Fukushima,Shugo Yajima,Wei Chen,Akihiro Hirakawa,Kenji Tanabe,Motohiro Fujiwara,Yuki Arita,Hajime Tanaka,Hitoshi Masuda,Yasuhisa Fujii & Soichiro Yoshida
Targeted Oncology Published:23 January 2026
DOI:https://doi.org/10.1007/s11523-025-01194-w
Abstract
Background
Given that immune checkpoint inhibitor-based regimens frequently yield delayed separation and late plateaus, conventional hazard ratio analyses that assume proportional hazards may misstate true benefit.
Objective
We aimed to test the validity of the proportional hazards assumption in first-line metastatic clear cell renal cell carcinoma trials and to compare the immune checkpoint inhibitor-based regimens using restricted mean survival time.
Methods
We performed a systematic review and network meta-analysis of phase III randomized controlled trials of first-line treatment for metastatic clear cell renal cell carcinoma, including immune checkpoint inhibitor-tyrosine kinase inhibitor combinations or dual-immune checkpoint inhibitor regimens. Individual patient data were reconstructed from the Kaplan–Meier curves of overall survival and progression-free survival. The restricted mean survival time differences were estimated.
Results
Five trials (4206 patients; six treatment arms) were examined. Proportional hazards assumption was violated in 60% of both overall survival and progression-free survival comparisons. In the restricted mean survival time-based network meta-analysis of overall survival, immune checkpoint inhibitor-tyrosine kinase inhibitor combinations, especially Nivolumab + Cabozantinib, dominated at 12–48 months, whereas Ipilimumab + Nivolumab ranked highest beyond 48 months. In the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) favorable-risk subgroup, Avelumab + Axitinib showed a favorable long-term profile despite the lack of statistical significance. In IMDC intermediate/poor-risk, patterns mirrored the overall population. For progression-free survival, Pembrolizumab + Lenvatinib ranked best across IMDC subgroups. Limitations included the reliance on reconstructed data and heterogeneity across trials.
Conclusions
Given the frequent proportional hazards violations, hazard ratio-only syntheses are insufficient for modern immune checkpoint inhibitor-based regimens. In the restricted mean survival time-based network meta-analysis, Pembrolizumab + Lenvatinib delivered rapid disease control, and Ipilimumab + Nivolumab showed the greatest late survival advantage in IMDC intermediate/poor-risk.
PROSPERO registration number
CRD420251143602.