2026-04-07 中国科学院(CAS)
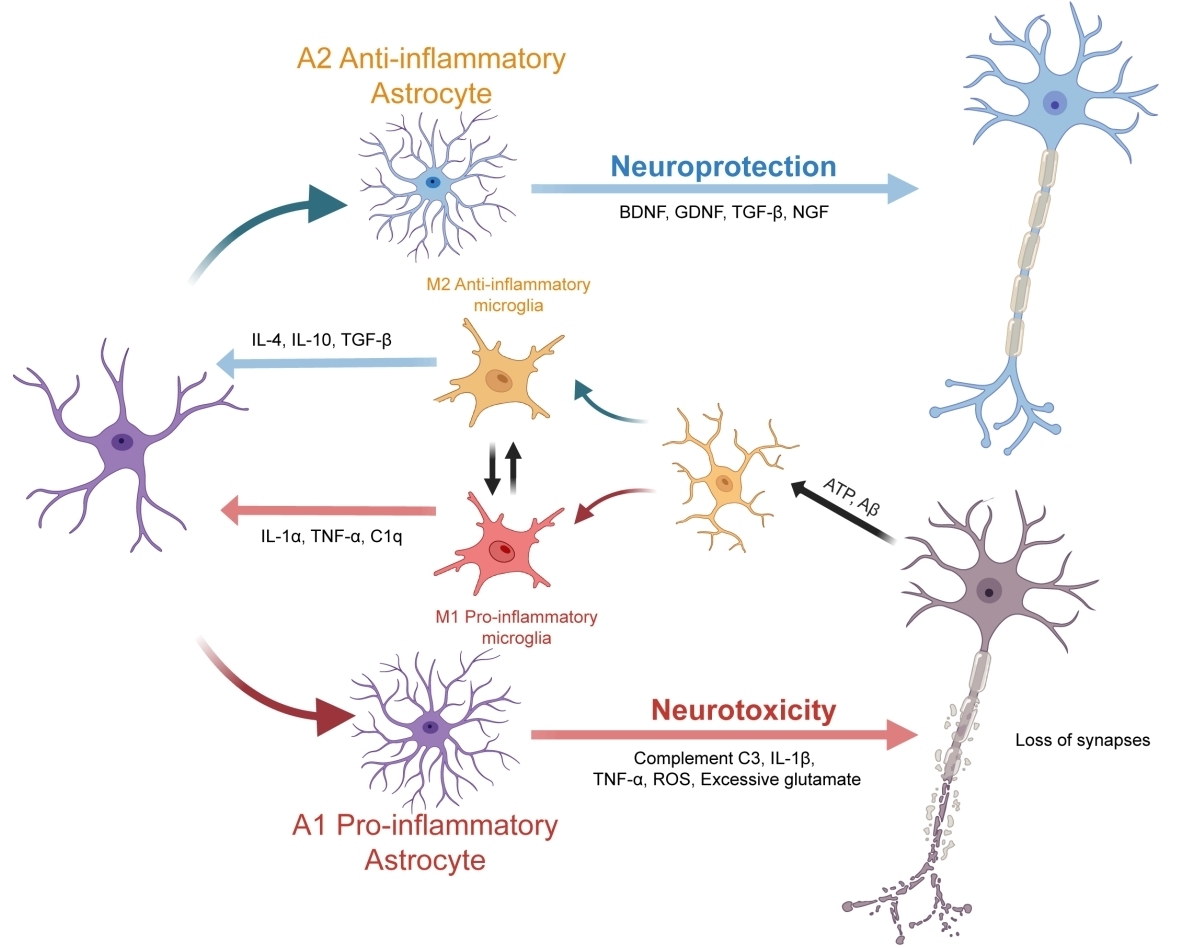
Molecular mechanisms of glial cell activation. (Image by SIAT)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202604/t20260408_1155366.shtml
- https://www.sciencedirect.com/science/article/abs/pii/S1568163726001121
神経変性疾患および脳血管疾患におけるグリア細胞の代謝および炎症における役割 Metabolic and inflammatory roles of glial cells in neurodegenerative and cerebrovascular diseases
Femi Abiola Ogunleye, Yumei Zhou, Lan Yan, Peace Osebhue Abhulimen, Yifei Xu, Mitchell K.P. Lai, Wahab Oluwanisola Okunowo, Gautam Sethi, Cheng Fang, Yinzhong Ma
Aging Research Reviews Available online: 2 April 2026
DOI:https://doi.org/10.1016/j.arr.2026.103120
Highlights
- Glial dysfunction fuels chronic neuroinflammation in neurodegenerative disorders.
- Activated microglia reprogram metabolism to glycolysis, producing ROS and pro-inflammatory cytokines.
- Astrocytic lipid dysregulation disrupts cholesterol transport, driving oxidative stress and neuronal loss.
- NF-κB, NLRP3 inflammasome, and complement C3/C3aR axis orchestrate persistent glial activation.
- Therapeutic targeting of glial metabolism and signaling shows promise for neuroprotection.
Abstract
Chronic neuroinflammation is a hallmark of neurodegenerative and cerebrovascular diseases and is largely driven by dysfunctional activation of microglia and astrocytes. Recent advances in single-cell transcriptomics and metabolic profiling have revealed the remarkable heterogeneity and plasticity of these glial cells, highlighting their dual roles in neuroprotection and neurotoxicity. Upon activation, microglia adopt pro-inflammatory phenotypes and undergo metabolic reprogramming characterized by a shift from oxidative phosphorylation to glycolysis, resulting in the release of cytokines and reactive oxygen species (ROS). Concurrently, astrocytes display dysregulated lipid metabolism, leading to the accumulation of toxic lipid species and amplification of inflammatory responses through metabolic and cytokine-mediated crosstalk, including lactate-mediated interactions between glial cells. Key inflammatory pathways regulate these processes, such as nuclear factor kappa B (NF-κB) signaling, activation of the NOD-, LRR-, and pyrin domain-containing protein 3 (NLRP3) inflammasome, and the complement component 3/complement component 3a receptor (C3/C3aR) signaling axis. These pathways coordinate glial activation and sustain deleterious feedback loops between microglia and astrocytes, ultimately promoting neuronal injury. These responses are often initiated by ROS and damage-associated molecular patterns (DAMPs) that trigger innate immune signaling. These pathological interactions are further amplified by impaired cholesterol transport, extracellular vesicle-mediated communication, and mitochondrial dysfunction. Collectively, these mechanisms exacerbate neuroinflammation and contribute to neurodegenerative progression and neurovascular impairment. Despite these advances, important gaps remain in understanding context-specific glial interactions and the limitations of current experimental models. This review summarizes current insights into glial-mediated neuroinflammation in ageing-related neurological disorders and discusses emerging therapeutic strategies targeting glial metabolism and immune signaling to mitigate disease progression.