2026-04-17 国立がん研究センター,名古屋大学,京都大学
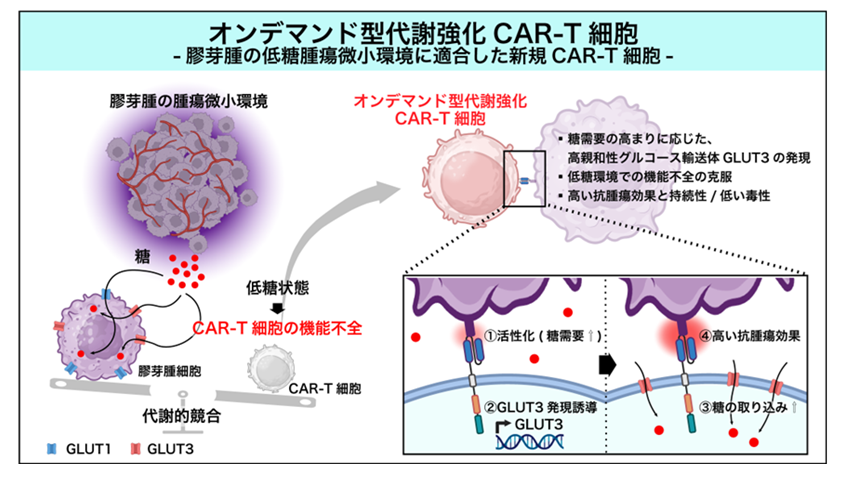
概要図
<関連情報>
- https://www.ncc.go.jp/jp/information/researchtopics/2026/0417/index.html
- https://www.science.org/doi/10.1126/scitranslmed.adu3532
オンデマンドGLUT3発現は、膠芽腫の前臨床モデルにおいてCAR T細胞の代謝能と抗腫瘍効果を高める On-demand GLUT3 expression augments CAR T cell metabolic fitness and antitumor efficacy in preclinical models of glioblastoma
Junya Yamaguchi, Keisuke Watanabe, Akihiro Nakamura, Nina Yi-Tzu Lin, […] , and Hiroyoshi Nishikawa
Science Translational Medicine Published:15 Apr 2026
DOI:https://doi.org/10.1126/scitranslmed.adu3532
Editor’s summary
Chimeric antigen receptor T cells (CAR T cells) are an attractive option to treat aggressive solid tumors, such as glioblastoma (GBM). However, the immunosuppressive and glucose-poor GBM microenvironment put CAR T cells at a disadvantage, given that their lower-affinity glucose transporter 1 (GLUT1) cannot outcompete the higher-affinity GLUT3 expressed by tumor cells. Here, Yamaguchi et al. engineered CAR T cells to express GLUT3; whereas systemic expression resulted in toxicity, an on-demand system controlled by CAR T cell activation boosted the activity of the CAR T cells, enabling tumor control without toxicity. These data highlight both the promise of CAR T cells for GBM and offer a technological advance to tailoring CAR T cell function. —Courtney Malo
Abstract
The clinical success of chimeric antigen receptor T cell (CAR T cell) therapy in hematologic malignancies has prompted its application for refractory solid tumors, including glioblastoma (GBM). However, CAR T cell trials against solid tumors have failed to show clinical efficacy thus far. Here, we show that the dysfunction of CAR T cells in GBM is attributed, at least, in part, to glucose deficiency in the tumor microenvironment (TME) driven by the substantial consumption of glucose by cancer cells. Engineering CAR T cells to continuously express glucose transporter 3 (GLUT3), a high-affinity glucose transporter, restored their cytokine production and killing activity. However, although CAR T cells with stable GLUT3 expression induced tumor reduction in a preclinical GBM model, their overactivation led to adverse events and mouse death. In contrast, on-demand GLUT3-expressing CAR T cells, in which GLUT3 transcription was driven by the nuclear translocation of nuclear factor of activated T cells (NFAT) as a consequence of target antigen stimulation, exhibited enhanced metabolic fitness and increased antitumor efficacy, leading to long-lasting tumor control in intracranial human GBM cell xenograft models while preventing adverse events. We propose that on-demand enhancement of metabolic fitness, such as at the time of exposure to tumor antigens, is a concept for boosting the antitumor efficacy of CAR T cells against solid tumors.

.jpg)