2026-04-27 東京科学大学
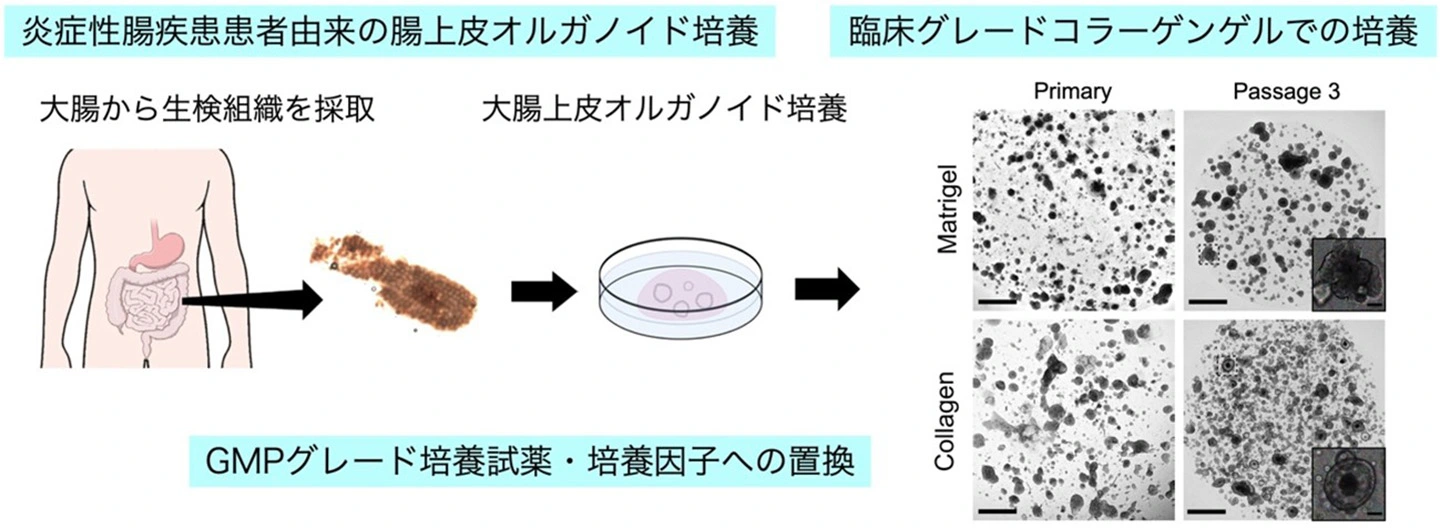
図1. 臨床応用を目指した患者由来ヒト大腸上皮オルガノイド培養法の開発
<関連情報>
- https://www.isct.ac.jp/ja/news/14o4hvxct60c
- https://link.springer.com/article/10.1186/s13287-026-04995-6
ペプチドベースのWntシグナル活性化により、再生細胞療法のための臨床グレードの患者由来腸管オルガノイドの大規模生産が可能になる Peptide-based Wnt signal activation enables scalable production of clinical-grade patient-derived intestinal organoids for regenerative cell therapy
Hady Yuki Sugihara,Sayaka Nagata,Sho Kawasaki,Junichi Takahashi,Yui Hiraguri,Masayoshi Fukuda,Kohei Suzuki,Tatsuro Murano,Satoru Fujii,Toshimitsu Fujii,Hiromichi Shimizu,Kazuo Ohtsuka,Mamoru Watanabe,Ryuichi Okamoto & Tomohiro Mizutani
Stem Cell Research & Therapy Published:29 March 2026
DOI:https://doi.org/10.1186/s13287-026-04995-6 Unedited version
Abstract
Background
Gastrointestinal diseases often involve cellular damage, degeneration or dysfunction in the tract, frequently requiring surgical interventions risking complications and lowered quality of life. Regenerative medicine holds great promise in improving patient care and providing novel treatment options for previously irreparable and untreatable tissues. Despite the clinical potential of intestinal organoids as a resource for regenerative cell therapy and bioengineering, the lack of clinical-grade cultures has hampered further development. Moreover, strategies to efficiently and reliably expand clinical-grade cultures at the scale required for application is limited.
Methods
A GMP-compliant protocol was developed to generate patient-derived colonic organoids from endoscopic biopsies. Clinical-grade colonic organoids cultured and expanded in Type-I collagen were compared to conventional Matrigel cultured organoids. To improve the culture-, cost-, and time-efficiency of culture expansion, several strategies were developed including organoid area-based passaging, one well plate culture, and the incorporation of Wnt activating peptide, PG-008. Conventional recombinant WNT3A culture was compared to the peptide PG-008 culture using single cell RNA sequencing.
Results
Clinical-grade collagen cultured organoids exhibited similar culture efficiency to Matrigel. Organoid establishment rate from 60 patients using the GMP-compliant protocol was 82%. The incorporation of PG-008 significantly enhanced organoid growth and stabilized patient-patient variability through intestinal stem cell (ISC) enrichment. Single cell RNA sequencing revealed that PG-008 resulted in remarkably pure culture consisting of ISCs and transit amplifying cells, suitable for rapid and consistent expansion. Intriguingly, our GMP-grade colonic organoids contained LGR5+ ISCs, and injury-induced LGR5− regenerative ISCs, both enriched in peptide culture.
Conclusions
Our study establishes clinical-grade colonic organoids for further application, including autologous transplantations and bioengineering. Further, collagen cultured organoids can be a valuable model facilitating in vitro investigations into regenerative stem cell induction and intestinal regeneration.