2026-02-27 産業技術総合研究所
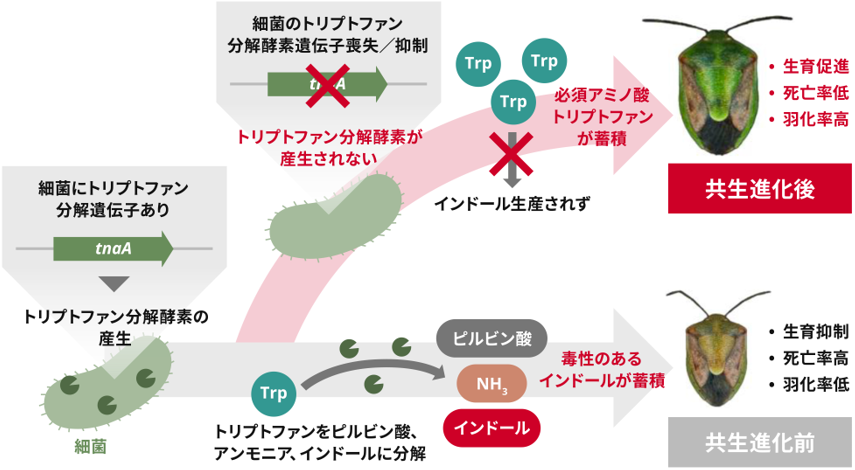
本研究で明らかになった大腸菌の共生細菌化の仕組み
<関連情報>
- https://www.aist.go.jp/aist_j/press_release/pr2026/pr20260227/pr20260227.html
- https://www.nature.com/articles/s41564-026-02264-z
トリプトファナーゼの破壊は昆虫と細菌の共生を促進する Tryptophanase disruption promotes insect–bacterium mutualism
Yayun Wang,Minoru Moriyama,Ryuichi Koga,Kohei Oguchi,Takahiro Hosokawa,Hiroki Takai,Shuji Shigenobu,Naruo Nikoh &Takema Fukatsu
Nature Microbiology Published:27 February 2026
DOI:https://doi.org/10.1038/s41564-026-02264-z
Abstract
Animal–microorganism symbioses are omnipresent, with both partners often gaining benefits as mutualists. A single mutation in the carbon catabolite repression system in Escherichia coli enables mutualism with the stinkbug Plautia stali. Here we find that this mutation is not present in natural symbioses. Given that the carbon catabolite repression pathway affects the expression of >500 downstream genes, we investigated their role in mutualisms. We find that disruption of a single gene, tnaA, encoding tryptophanase makes E. coli mutualistic to P. stali, resulting in the accumulation of tryptophan and the reduction of toxic indole. A survey of wild populations of P. stali and other stinkbug species revealed that their typical microbial symbionts, Pantoea, consistently lack the tnaA gene. Some Pantoea species such as Pantoea ananatis retain the tnaA gene and cannot establish symbiosis with P. stali, but tnaA-disrupted P. ananatis partially restored the symbiotic capability. When a natural Pantoea mutualist of P. stali was transformed with a functional tna operon, its symbiotic capability reduced significantly. Our finding suggests that tryptophanase disruption may have facilitated the evolution of gut bacterial mutualists in insects.