2026-03-30 バッファロー大学(UB)
<関連情報>
- https://www.buffalo.edu/news/releases/2026/03/fat-molecule-can-help-trigger-both-cell-limbo-and-cell-death.html
- https://www.sciencedirect.com/science/article/abs/pii/S2451945626000735
ER局在性セラミド蓄積は複製老化に寄与する ER-localized ceramide accumulation contributes to replicative senescence
Shweta Chitkara, Mengru Li, Natasha Gozali, Andrey Kuzmin, Artem Pliss, Paras Prasad, Yasemin Sancak, G. Ekin Atilla-Gokcumen
Cell Chemical Biology Available online: 30 March 2026
DOI:https://doi.org/10.1016/j.chembiol.2026.03.003
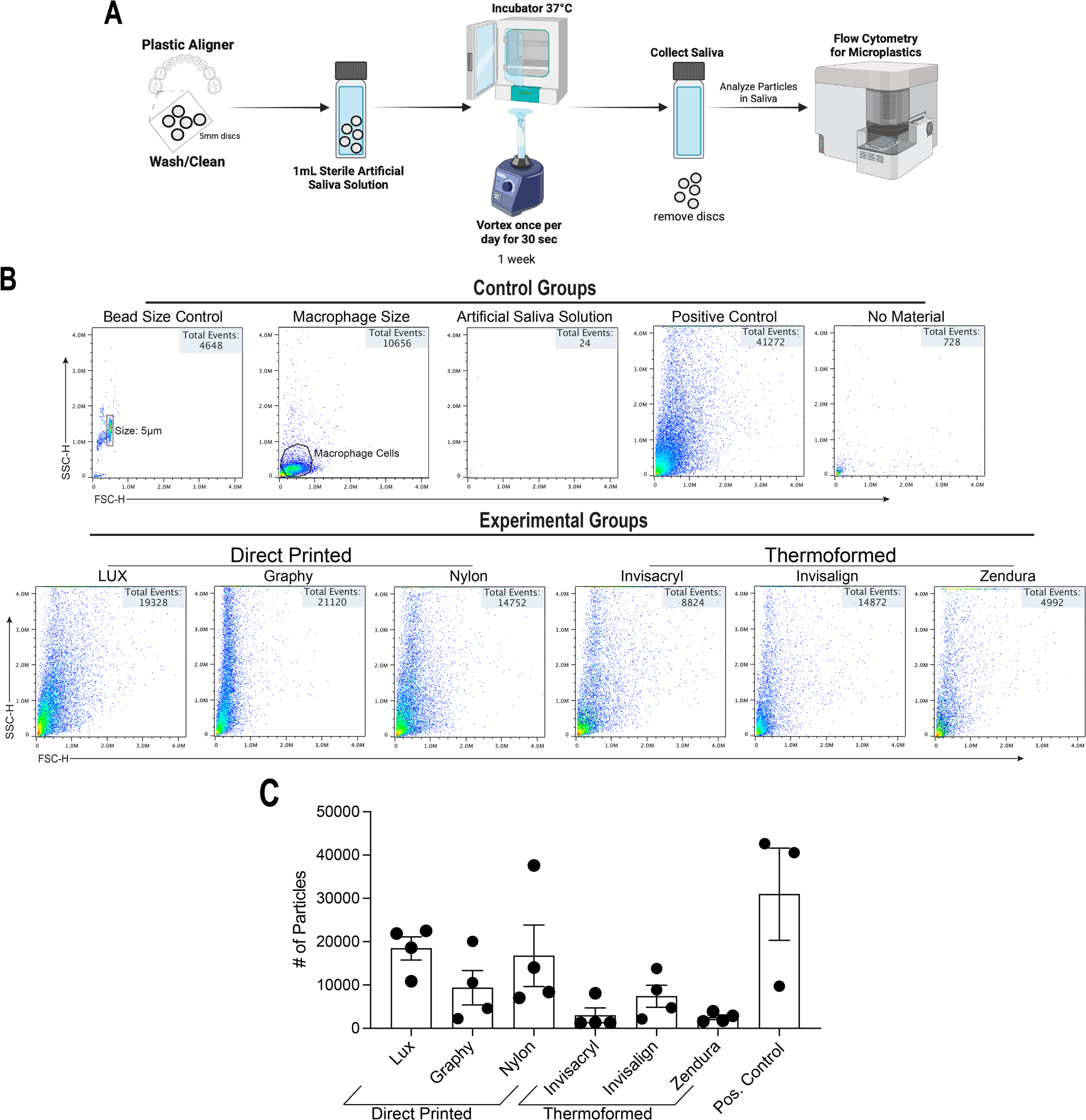
Graphical abstract

Highlights
- Replicative senescence features ceramide accumulation with depletion of sphingomyelins
- Sphingomyelin depletion despite unchanged SMS activity suggests impaired ceramide flux
- CERT inhibition phenocopies senescence
- Ceramides accumulate at the ER in senescent cells
Significance
Lipids function as spatially regulated signaling molecules. Their biological activities can depend on their subcellular localization as much as their abundance. Ceramides exemplify this principle: mitochondrial ceramide accumulation promotes apoptosis, yet how ceramide compartmentalization regulates alternative cell fate programs has remained unclear. Here, we establish that replicative senescence is defined not only by ceramide elevation, but also by disrupted ceramide trafficking that drives its accumulation at the endoplasmic reticulum (ER). We identify impaired ER-to-Golgi ceramide transport via CERT as a key regulatory node underlying senescence-associated sphingolipid remodeling. Functionally, perturbing ceramide trafficking phenocopies the lipid and phenotypic features of senescence, revealing lipid transport as a determinant of cell fate. By linking ER-localized ceramide accumulation to ER stress signaling, this work uncovers a connection between lipid spatial organization and senescence programming, highlighting organelle-resolved lipid metabolism as a targetable axis in aging biology.
Summary
Ceramides regulate diverse cellular processes through compartment-specific accumulation. While mitochondrial ceramide accumulation promotes apoptosis, its regulation and function during senescence remain incompletely understood. Here, we integrate lipidomics, transcriptomics, Raman spectroscopy, and biochemical characterizations to define sphingolipid remodeling in replicative senescence. Senescent cells exhibit elevated ceramide levels and depletion of very-long-chain sphingomyelins, despite unaltered sphingomyelin synthase 1 expression, implicating impaired ceramide-sphingomyelin turnover. Pharmacological inhibition of ceramide transfer protein (CERT), the ER-to-Golgi ceramide transporter, phenocopies sphingolipid remodeling and enhances senescence, suggesting disrupted ceramide trafficking as a driver of senescence. Raman spectroscopy suggests ceramide accumulation localized to the ER. In parallel, analysis of ER-enriched fractions confirms increased ceramide levels in ER fractions of senescent cells. Mechanistically, ceramide accumulation at the ER can contribute to ER stress. These findings identify altered ceramide trafficking as a contributor to ER stress and highlight ER-localized ceramide as a critical component of senescence-associated sphingolipid remodeling.