2026-03-31 東京大学, 順天堂大学, 群馬大学
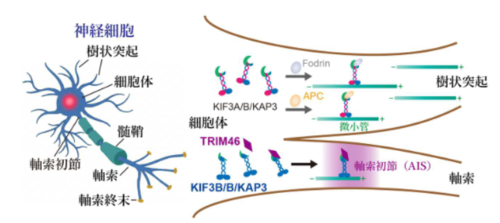
キネシン‐2複合体のサブタイプ分化によるTRIM46の軸索初節(AIS)への選択的輸送モデル 神経細胞において、KIF3A/B/KAP3からなるキネシン‐2複合体が構成の違いによって機能分担し、TRIM46をAIS へ特異的に輸送・集積させる模式図。
<関連情報>
- https://www.u-tokyo.ac.jp/content/400283813.pdf
- https://rupress.org/jcb/article-abstract/225/5/e202503138/281711/The-KIF3B-B-KAP3-tail-domain-specifically?redirectedFrom=fulltext
KIF3B/B/KAP3の尾部ドメインは、TRIM46の軸索起始部への輸送を特異的に促進する
The KIF3B/B/KAP3 tail domain specifically facilitates TRIM46 transport to the axon initial segment
Xuguang Jiang,Sotaro Ichinose,Tadayuki Ogawa,Kento Yonezawa,Nobutaka Shimizu,Nobutaka Hirokawa
Journal of Cell Biology Published:March 30 2026
DOI:https://doi.org/10.1083/jcb.202503138
Intracellular transport is essential for neuronal organization, yet how motor proteins achieve cargo selectivity remains incompletely understood. Kinesin-2 motors transport diverse cargos through the heterotrimeric KIF3/KAP3 complex, but whether variations in assembly composition contribute to functional specificity has been unclear. This study provides evidence for heterogeneity in neuronal KIF3/KAP3 assemblies, including a KIF3B-enriched, KAP3-associated population in addition to the canonical KIF3A/B/KAP3 complex. Biochemical and cellular analyses support a preferential association between this KIF3B-enriched assembly and TRIM46, a protein required for axon initial segment organization. Structural analyses further suggest that differences in tail conformation accompany distinct assembly states and may underlie cargo selectivity. Together, these findings support a model in which compositional and structural diversity within kinesin-2 complexes contributes to spatially regulated transport during neuronal development.