2026-04-23 カーディフ大学
<関連情報>
- https://www.cardiff.ac.uk/news/view/3042058-covid-antiviral-speeds-recovery-but-doesnt-reduce-hospitalisation-in-vaccinated-patients
- https://www.nejm.org/doi/full/10.1056/NEJMoa2502457
高リスク外来患者におけるCOVID-19に対する経口ニルマトレルビル・リトナビル併用療法 Oral Nirmatrelvir–Ritonavir for Covid-19 in Higher-Risk Outpatients New England Journal of Medicine
Christopher C. Butler, F.Med.Sci., Andrew D. Pinto, M.D., Victoria Harris, Ph.D., Jane Holmes, Ph.D., Najib M. Rahman, D.Phil., Lucy Cureton, B.Sc., Gail Hayward, D.Phil., +35 , for the PANORAMIC Trial and CanTreatCOVID Trial Collaborative Groups
The New England Journal of Medicine Published: April 22, 2026
DOI:10.1056/NEJMoa2502457
Abstract
Background
Nirmatrelvir–ritonavir has been shown to reduce progression to severe illness from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in unvaccinated high-risk outpatients. The effectiveness of nirmatrelvir–ritonavir in persons who have been vaccinated, infected naturally, or both is unclear.
Methods
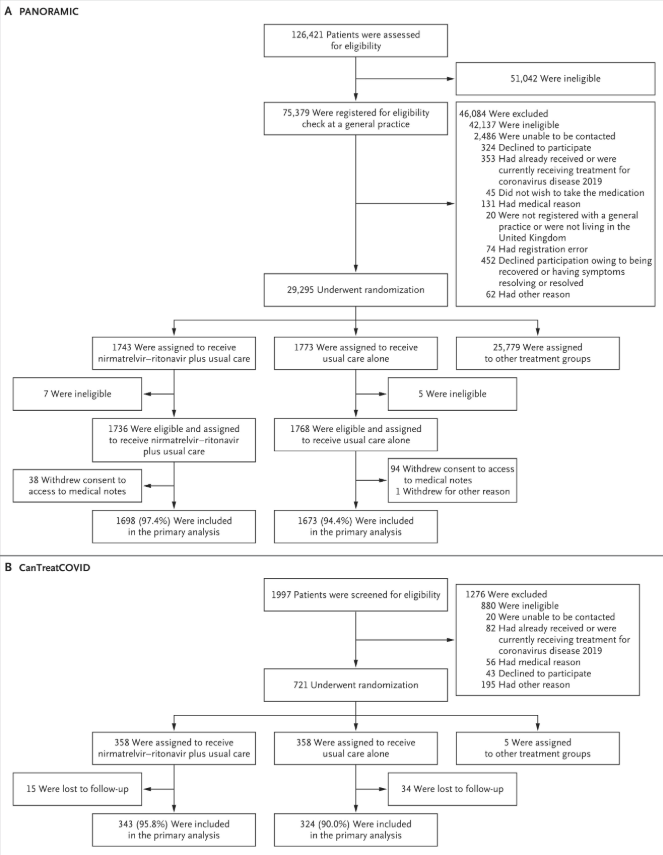
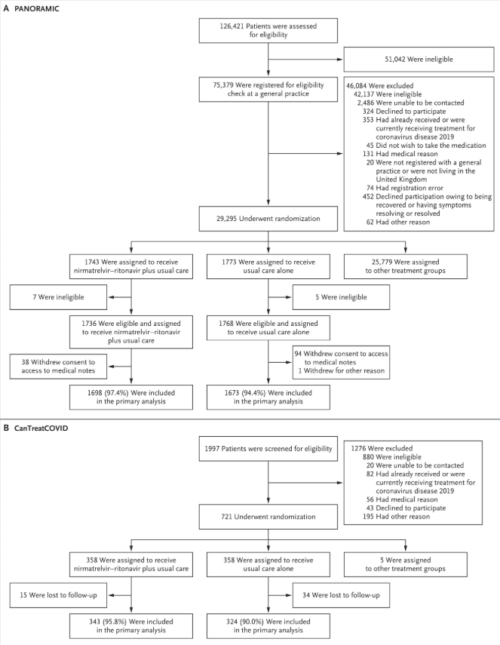
In two open-label platform trials (PANORAMIC in the United Kingdom and CanTreatCOVID in Canada), we enrolled higher-risk adults (≥50 years of age or ≥18 years of age with coexisting conditions) in the community who tested positive for SARS-CoV-2 and had been unwell for 5 days or less. The participants were randomly assigned to receive usual care plus nirmatrelvir (300 mg)–ritonavir (100 mg) twice a day for 5 days or to receive usual care alone. The primary outcome was hospitalization or death from any cause within 28 days after randomization.
Results
From December 8, 2021, to September 30, 2024, a total of 3516 participants in the PANORAMIC trial and 716 participants in the CanTreatCOVID trial underwent randomization. In the PANORAMIC trial, 14 of 1698 participants (0.8%) in the nirmatrelvir–ritonavir group and 11 of 1673 participants (0.7%) in the usual-care group were hospitalized or died (adjusted odds ratio, 1.18; 95% Bayesian credible interval, 0.55 to 2.62; probability of superiority, 0.334). In the CanTreatCOVID trial, 2 of 343 participants (0.6%) in the nirmatrelvir–ritonavir group and 4 of 324 participants (1.2%) in the usual-care group were hospitalized or died (adjusted odds ratio, 0.48; 95% Bayesian credible interval, 0.08 to 2.23; probability of superiority, 0.830). In a substudy involving 634 participants, viral load was reduced by the end of treatment with nirmatrelvir–ritonavir. Serious adverse events with nirmatrelvir–ritonavir were reported in 9 participants in the PANORAMIC trial and in 4 participants in the CanTreatCOVID trial.
Conclusions
In two open-label trials, nirmatrelvir–ritonavir did not reduce the incidence of hospitalization or death among vaccinated higher-risk participants with SARS-CoV-2 infection. (Funded by the National Institute for Health and Care Research, and others; PANORAMIC ISRCTN number, 2021-005748-31; CanTreatCOVID ClinicalTrials.gov number, NCT05614349.)