2026-04-27 カリフォルニア大学アーバイン校(UCI)
<関連情報>
- https://news.uci.edu/2026/04/27/forever-chemicals-may-be-linked-to-childhood-leukemia/
- https://www.nature.com/articles/s41370-026-00891-6
- https://medibio.tiisys.com/149463/
新生児乾燥血液スポット中のパーフルオロアルキル物質およびポリフルオロアルキル物質の標的分析および非標的分析と小児急性リンパ性白血病のリスク Targeted and non-targeted analyses of per-and polyfluoroalkyl substances in newborn dried blood spots and risk of childhood acute lymphoblastic leukemia
Veronica M. Vieira,Sheng Liu,Libby M. Morimoto,Jeremy Koelmel,Natalie R. Binczewski,Joseph L. Wiemels,Xiaomei Ma,Krystal J. Godri Pollitt & Catherine Metayer
Journal of Exposure Science & Environmental Epidemiology Published:14 April 2026
DOI:https://doi.org/10.1038/s41370-026-00891-6
Abstract
Background
Per- and polyfluoroalkyl substances (PFAS) have carcinogenic potential but are understudied in relation to childhood cancers.
Objective
We examined associations between targeted and non-targeted PFAS measured in newborn dried blood spots (DBS) and the risk of childhood acute lymphoblastic leukemia (ALL) in Los Angeles County, California, accounting for maternal and child characteristics.
Methods
ALL cases (n = 125) diagnosed before age 18 years during 2000–2015 and controls (n = 219) were selected from a registry-based study using stratified sampling based on birth year and birth address within a PFAS-contaminated water district according to the USEPA Third Unregulated Contaminant Monitoring Rule. We calculated design-based odds ratios (OR) and 95% confidence intervals (CI) for the effect of PFAS exposures, independently and adjusting for other PFAS. We also conducted non-linear and stratified analyses.
Results
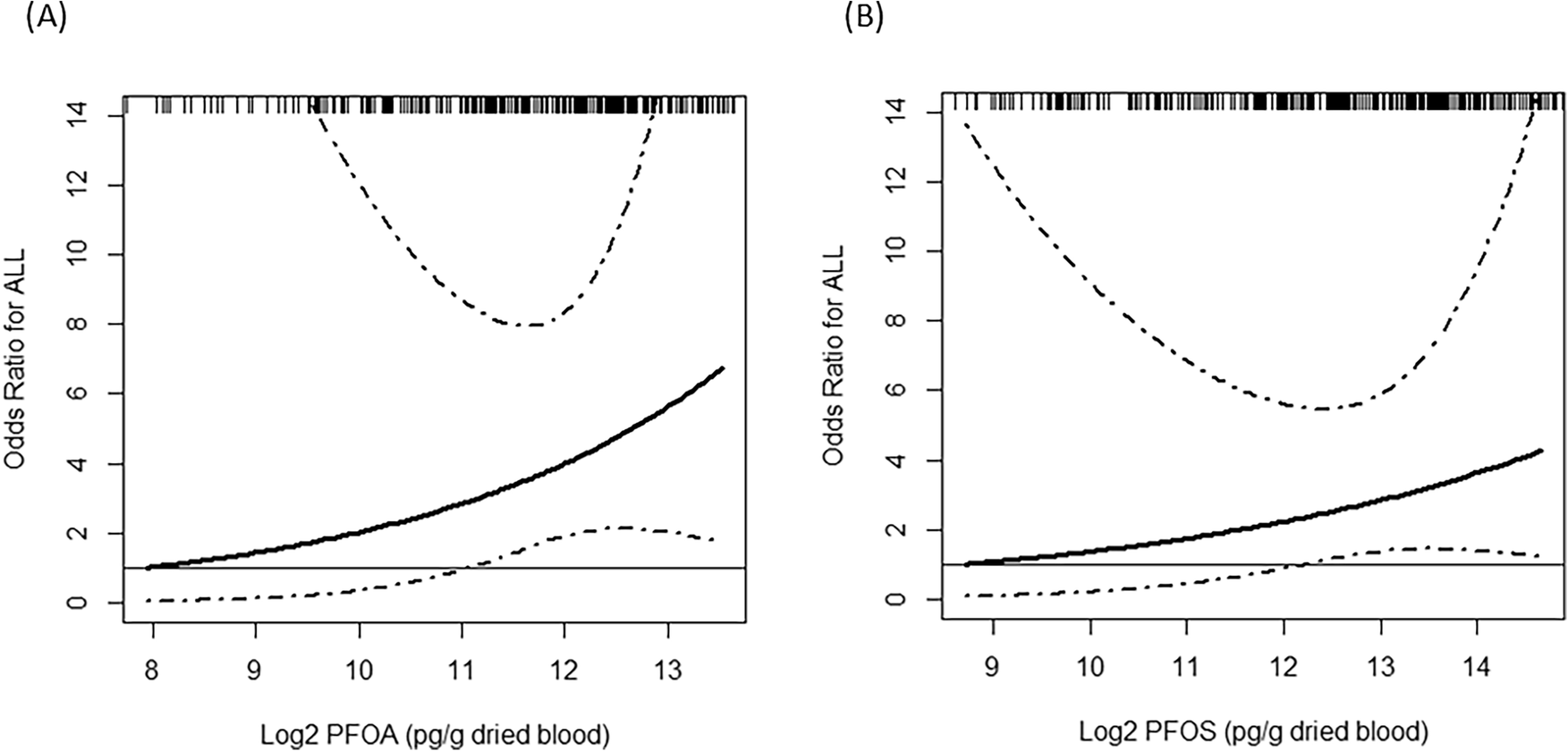
Of the 17 PFAS quantified using targeted analysis, perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) had the highest mean concentrations in DBS, with 4690 and 10,307 pg/g dried blood among cases compared to 4245 and 8142 pg/g dried blood among controls, respectively. The highest risks were observed for the 4th exposure quartile compared with the 1st quartile (PFOA OR = 1.56, CI: 0.42, 5.73; PFOS OR = 1.64, CI: 0.44, 6.14). In non-linear statistical analyses of joint PFOA and PFOS exposures adjusted for other detected PFAS, we also found that ALL risk increased with increasing levels of log2-PFOA and log2-PFOS. Non-targeted analysis identified 26 additional PFAS, for which elevated risk of childhood ALL was associated with a doubling of C4HF7O3 exposure (OR = 5.04, CI: 1.08, 23.63) and the highest quartile of C10HF19O5 exposure (OR = 5.20, CI: 1.15, 23.56). Associations were generally stronger among non-Hispanic participants compared to Hispanic participants, but these analyses were limited by small sample sizes and should be considered exploratory.
Significance
There was some suggestion that high PFOA and PFOS exposures measured at birth, as well as certain PFAS detected by non-targeted approaches, were related to childhood ALL risk.
Impact statement
This study of childhood ALL in Los Angeles County, California found increased risk among children with the highest PFOA and PFOS levels measured in neonatal dried blood spots. These risks were stronger among non-Hispanic children, and the greatest risk was observed for joint PFOA and PFOS exposure adjusting for other detected PFAS and maternal/child characteristics. We also observed possible associations with PFAS discovered by non-targeted analysis. Our results highlight the utility of dried blood spots as a matrix for assessing early life exposures as well as the value of hybrid targeted and non-targeted approaches to measure PFAS in health studies.

