2026-02-27 上海交通大学(SJTU)
<関連情報>
- https://global.sjtu.edu.cn/en/news-events/news/2465
- https://www.nature.com/articles/s41586-026-10113-6
Chd3の生体内塩基編集はマウスの行動異常を救済する In vivo base editing of Chd3 rescues behavioural abnormalities in mice
Kan Yang (杨侃),Wei-Ke Li (李维克),Yi-Xiao Geng (耿一啸),Shu-Qian Zhang (张淑倩),Shi-Hao Wu (吴诗昊),Yan-Bo Cheng (程艳波),Jun-Wen Wang (王俊文),Zhan-Kui Xu (许占魁),Wen-Xin Wang (汪闻欣),Tan-Ying Zhang (张谭颖),Pei-Ye Wang (王培烨),Yi-Ting Yuan (袁依婷),Juan Fan (范娟),Jun Wu (吴君),Ruo-Chuan Xu (徐若川),Yue-Fang Zhang (张月芳),Gong-Jia Tao (陶宫佳),Zheng-Hui Li (李郑晖),Chen-Xi Lin (林晨曦),Tian-Shu Li (李天舒),Xin-Yi Zhang (张心怡),Jie Li (李洁),Ru Zhang (张儒),Wen-Xiu Yang (杨文秀),… Zilong Qiu (仇子龙)
Nature Published:18 February 2026
DOI:https://doi.org/10.1038/s41586-026-10113-6
Abstract
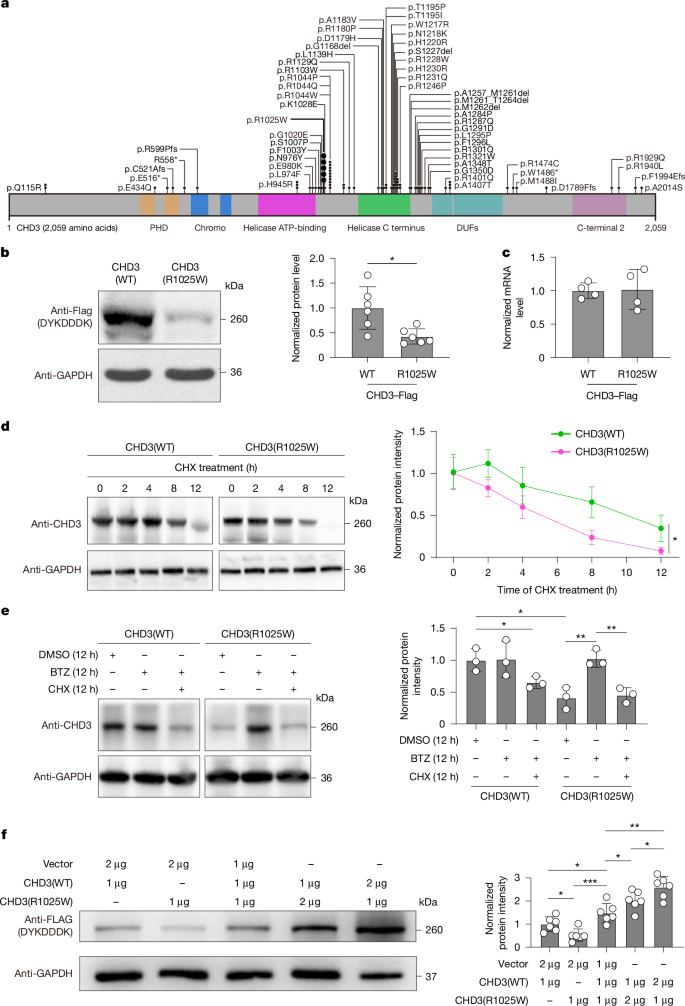
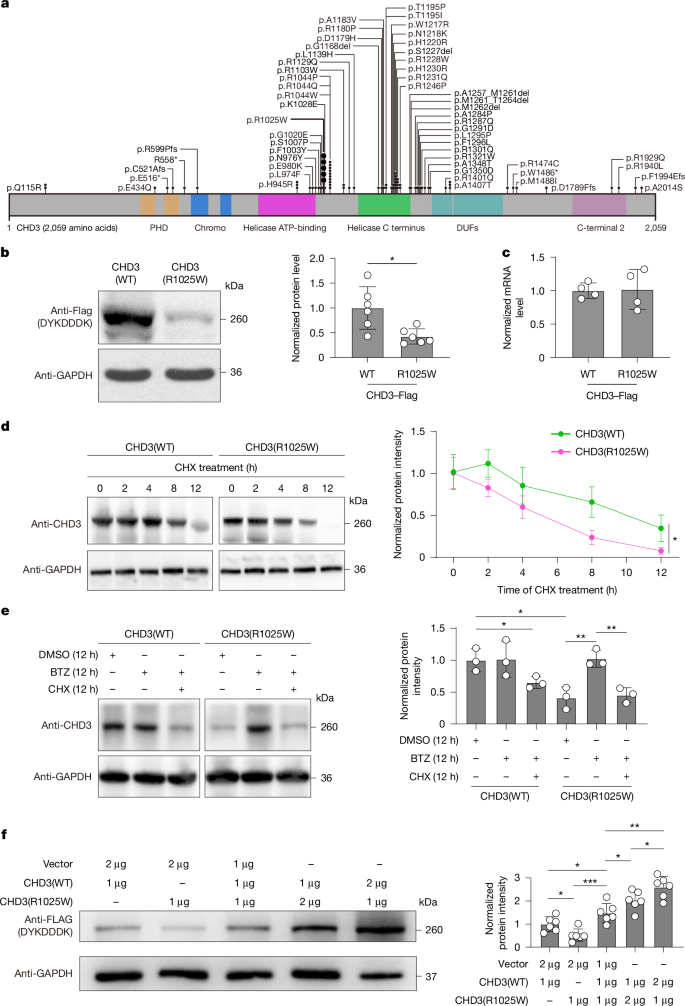
Neurodevelopmental disorders that arise from de novo mutations in chromatin-remodelling genes lack targeted treatments. Snijders Blok–Campeau syndrome (SNIBCPS)1, which is caused by pathogenic variants in CHD3, manifests with intellectual disability, autistic-like behaviours and motor deficits2. Whether somatic gene correction can reverse such phenotypes in vivo remains unknown. Here we show that modelling the recurrent CHD3 variant p.R1025W in a humanized mouse model (Chd3hR1025W/+) recapitulates key features of SNIBCPS, including reduced CHD3 protein levels and abnormalities in social communication, cognition and motor coordination. We engineered a TadA-embedded adenine base editor (TeABE) and delivered it brain-wide using a dual adeno-associated virus (AAV) system and achieved efficient on-target A•T-to-G•C correction across multiple cortical and hippocampal regions with minimal bystander activity. This intervention restored CHD3 levels and ameliorated behavioural abnormalities in vivo. Furthermore, intrathecal dual AAV delivery in nonhuman primates resulted in widespread neuronal transduction and efficient TeABE reconstitution, a result that supports its translational feasibility. These findings establish in vivo base editing as a viable therapeutic approach for CHD3-related neurodevelopmental disease. More broadly, they demonstrate that precise single-base correction in the postnatal brain can restore protein dosage and function, thereby offering a framework for the treatment of monogenic neurodevelopmental disorders.