2026-03-31 東京科学大学
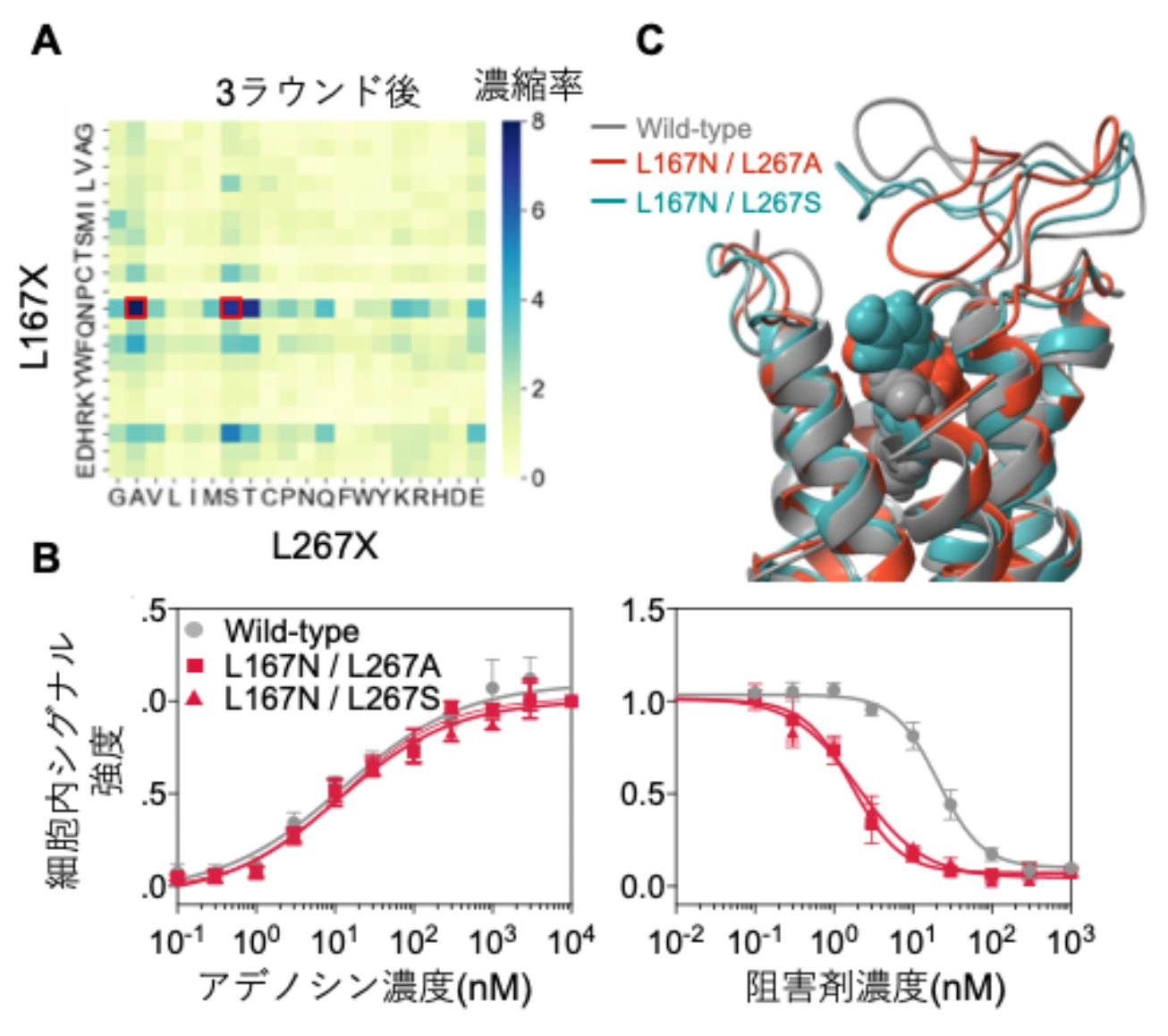
図1. 人工進化により得られたA2AR変異体。
<関連情報>
リボソームディスプレイを用いた拮抗薬結合に基づくアデノシンA2A受容体の試験管内進化 In Vitro Evolution of the Adenosine A2A Receptor Based on an Antagonist Binding Using a Ribosome Display
Genki Fukasawa,Yuma Matsuoka,Duy Phuoc Tran,Haruka Nishigaki,Keisuke Fukunaga,Takayoshi Watanabe,Tomohiro Doura,Naohiro Terasaka,Ako Kagawa,Takeshi Murata,Akio Kitao,Shigeki Kiyonaka,and Tomoaki Matsuura
Journal of the American Chemical Society Published: March 6, 2026
DOI:https://doi.org/10.1021/jacs.6c02372
Abstract
In vitro evolution is one of the powerful methods for altering the characteristics of a protein of interest; however, its application to membrane proteins remains challenging due to the requirement of a membrane environment in vitro. This study presents the first successful in vitro evolution of human adenosine A2A receptor (A2AR), one of the G protein-coupled receptors (GPCRs), achieved through combining cell-free protein synthesis (CFPS), a nanodisc for membrane mimicry, and a ribosome display. A saturation mutagenesis library of A2AR targeting the L167 and L267 residues, a library with a diversity of only 400, was constructed without sequence bias. Three rounds of affinity selection against A2AR-selective antagonist ZM241385 enriched the L167N/L267A and L167N/L267S mutants. These mutants exhibited greater than 10-fold improved sensitivity to ZM241385 in mammalian cell-based assays while retaining responsiveness to the endogenous ligand adenosine. Molecular dynamics simulations revealed new interactions between ZM241385 and A2AR mutants that are likely to contribute to stabilizing the conformation of the complexes. Finally, as an example of an application, the obtained mutant was used for cell-type-specific inhibition of A2AR signaling by ZM241385. This study demonstrates a powerful strategy for engineering GPCRs entirely in vitro with tailored ligand-binding properties, even from a library with small diversity, and shows that the evolved mutants exhibit improved functionality in mammalian cells.