2026-04-14 琉球大学
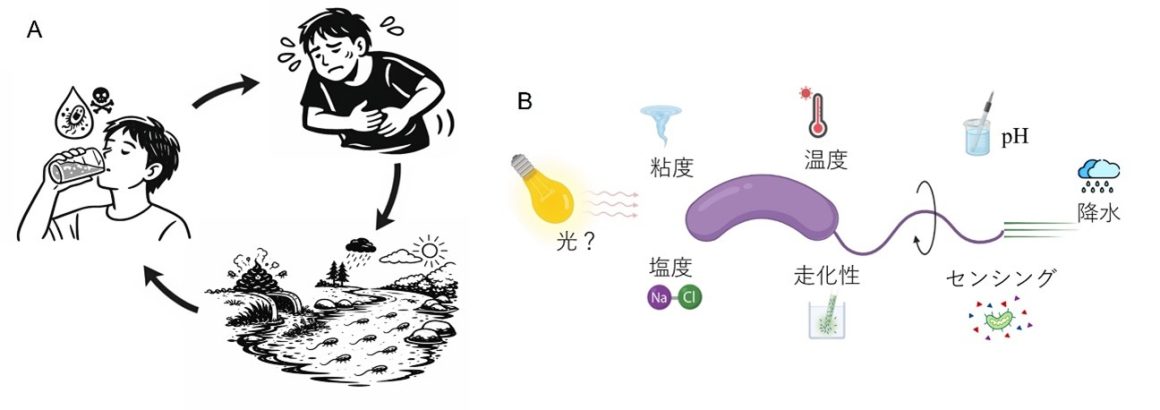
図1 コレラ菌の感染サイクルと環境応答
<関連情報>
光活性化cAMPシグナル伝達は、コレラ菌のナトリウム駆動運動を制御する Light-activated cAMP signaling controls sodium-driven motility in Vibrio cholerae
Jun Xu, Shuichi Nakamura, Suzuna Tomoyose, +2 , and Tetsu Yamashiro
Proceedings of the National Academy of Sciences Published:April 9, 2026
DOI:https://doi.org/10.1073/pnas.2530860123
Significance
Bacteria use second messengers to couple environmental cues to behavior, but how light regulates motility in Vibrio cholerae is not well defined. We show that in strain AJ10, a photoresponsive adenylyl cyclase (CyaA) elevates intracellular cAMP under illumination and is required for light-enhanced swimming. Light responsiveness persists under nutrient limitation, linking an ecologically relevant cue to cyclic-nucleotide signaling and sodium-powered flagellar energetics. These findings define a light–cAMP–motility axis that links environmental illumination to second messenger signaling and sodium-powered flagellar energetics in a major bacterial pathogen.
Abstract
Light is one of the most pervasive physical cues in aquatic environments, yet its impact on nonphototrophic pathogens remains largely unexplored. Here, we show that a strain of cholera bacterium Vibrio cholerae directly couples illumination to motility through cyclic AMP (cAMP) signaling. Exposure to visible light rapidly elevates intracellular cAMP and increases swimming speed, whereas deletion of the single adenylyl cyclase gene (cyaA) abolishes both responses; complementation or addition of exogenous cAMP restores the phenotype. Heterologous expression of V. cholerae CyaA in an Escherichia coli ΔcyaA ΔcpdA background reconstitutes light-activated cAMP synthesis, indicating that CyaA confers photoreactivity. Purified CyaA exhibits a reversible light-dependent spectral shift consistent with flavin-dependent photochemistry, identifying it as a light-responsive cyclase. Illumination triggers rapid membrane hyperpolarization and sodium efflux, strengthening the sodium-motive force that powers the flagellar motor. This response persists under nutrient-limited conditions. Together, these findings define a light → cAMP → sodium-motive force coupling axis in V. cholerae, suggesting that ambient light may influence motility and dispersal in sunlit environments.

