2026-04-15 岡山大学,大阪大学,愛媛県立医療技術大学,早稲田大学,米国アーカンソー大学,北海道大学,大阪公立大学
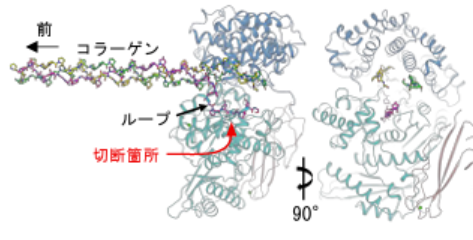
図1.細菌性コラゲナーゼは三重らせん型 コラーゲンをほぐしてα鎖(赤)を引き出す。
<関連情報>
- https://www.okayama-u.ac.jp/tp/release/release_id1533.html
- https://www.okayama-u.ac.jp/up_load_files/press_r8/press20260415-1.pdf
- https://www.nature.com/articles/s41467-026-71099-3
細菌性コラゲナーゼはコラーゲンの幾何学的構造を利用して連続的な切断を行う Bacterial collagenase harnesses collagen geometry for processive cleavage
Hiroya Oki,Katsuki Takebe,Adjoa Bonsu,Kazunori Fujii,Ryo Masuda,Nicholas Henderson,Takehiko Mima,Takaki Koide,Mahmoud Moradi,Osamu Matsushita,Joshua Sakon & Kazuki Kawahara
Nature Communications Published:02 April 2026
DOI:https://doi.org/10.1038/s41467-026-71099-3 Unedited version
Abstract
Collagen, the major structural protein in the animal extracellular matrix, forms a triple helix that resists proteolysis and requires specialised enzymes for degradation. Flesh-eating bacteria secrete collagenases that unwind the collagen triple helix and processively trim Gly–X–Y triplet repeats, yet the molecular basis of this process has remained obscure. Here, cryo-electron microscopy reveals how Hathewaya histolytica collagenase ColH engages its substrate and exploits the helix’s architecture for catalysis. ColH encircles a single collagen triple helix in a closed-ring conformation and, through dynamic domain motions, dehydrates and destabilises it. The enzyme undergoes substrate-assisted twisting to adopt a rigid ratcheted conformation, in which one chain is bent into a tripeptide-long ‘bight’ and threaded into the active site for cleavage, while two uncut strands are partitioned to non-catalytic sites. Release of the bight appears to reset the enzyme, with the uncut strands serving as guiding tracks. Repeated cycling between dynamic and rigid states likely enables triplet-by-triplet translocation, allowing ColH to harness collagen’s geometry for processive degradation. These findings reveal a bacterial strategy for collagen unwinding and cleavage distinct from that of mammalian collagenases, highlighting divergent evolutionary solutions for degrading one of nature’s most intractable substrates.