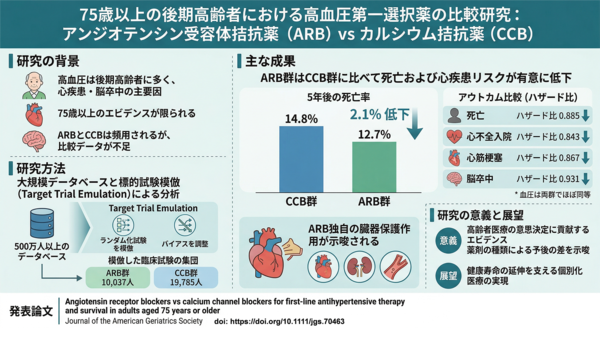
2026-04-27 理化学研究所,科学技術振興機構

図1 熱ストレスと傷害ストレスに共通して誘導される遺伝子の同定
公開RNA-seqデータの再解析により、傷害ストレス後12時間以内に発現上昇する5,615個の遺伝子のうち、1,932個の遺伝子が熱ストレスによっても誘導されることが明らかになった。
<関連情報>
- https://www.riken.jp/press/2026/20260427_1/index.html
- https://academic.oup.com/plcell/advance-article-abstract/doi/10.1093/plcell/koag124/8661150
傷害はHSFA1転写因子を活性化し、シロイヌナズナの細胞再プログラミングを促進する Wounding activates the HSFA1 transcription factors to promote cellular reprogramming in Arabidopsis
Duncan Coleman,Akira Iwase,Ayako Kawamura,Arika Takebayashi,Yu Chen,Max Minne,Katja E Jager,Maolin Peng,Yutaka Kodama,David S Favero,…
The Plant Cell Published:22 April 2026
DOI:https://doi.org/10.1093/plcell/koag124
Abstract
Mechanical injury is a primary trigger for cellular reprogramming during organ regeneration, yet the molecular mechanisms that link wounding to reprogramming remain poorly understood. In this study we identify the Arabidopsis HEAT SHOCK FACTOR A1 (HSFA1) class of transcription factors, which are key regulators of the heat stress response, as central players in wound-induced callus formation and shoot regeneration. Loss of HSFA1 function in the hsfa1abd triple or hsfa1abde quadruple mutants severely impairs cellular reprogramming, reducing callus formation from wounded hypocotyls, as well as shoot regeneration from explants. Conversely, overexpression of the HSFA1d gain-of-function variant markedly enhances regeneration. Time-series RNA-seq and ChIP-seq analyses revealed that HSFA1 directly activates the key reprogramming regulators WOUND-INDUCED DEDIFFERENTIATION 1 (WIND1), PLETHORA 3 (PLT3) and ZINC FINGER OF ARABIDOPSIS THALIANA 6 (ZAT6). Furthermore, we demonstrate that HSFA1d activity is attenuated by SAP AND MIZ1 DOMAIN- CONTAINING LIGASE1 (SIZ1)-mediated SMALL UBIQUITIN-LIKE MODIFIER (SUMO)ylation, linking post-translational modification to the regulation of wound responses. Our findings establish HSFA1 as an early transcriptional hub that integrates wound signals with the activation of a broad gene network that drives cellular reprogramming, thereby providing a mechanistic framework for understanding how stress-responsive transcription factors control regeneration.