2026-04-20 東北大学
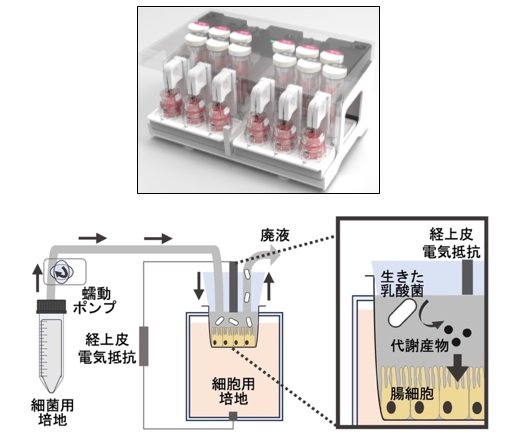
図1. 本研究で用いたマイクロ流体共培養デバイスの写真とその模式図。経上皮電気抵抗を継時的に測定することにより、細胞のタイトジャンクションを維持したまま、細菌と共培養できていることを確認することができる。
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/04/press20260420-02-Lactiplantibacillus.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20260420_02web_Lactiplantibacillus.pdf
- https://www.cell.com/iscience/fulltext/S2589-0042(26)00891-6
生菌および加熱処理したLactiplantibacillus plantarumは、腸管上皮細胞において異なる代謝反応および免疫反応を誘導する Live and heat-treated Lactiplantibacillus plantarum induce distinct metabolic and immune responses in intestinal epithelial cells
Kaho Matsumoto ∙ Yuta Takada ∙ Yoshiya Imamura ∙ … ∙ Toyoyuki Hashimoto ∙ Keita Nishiyama ∙ Haruki Kitazawa
iScience Published:March 31, 2026
DOI:https://doi.org/10.1016/j.isci.2026.115516
Highlights
- Compared host responses to live and heat-treated L. plantarum using microfluidics
- Live L. plantarum induces glycolytic shift and PPARG signaling in epithelia
- Heat-treated L. plantarum activates NFKB signaling via altered surface structures
- Bacterial viability shaped epithelial metabolic and immune responses in co-culture
Summary
Probiotics and postbiotics are distinguished by bacterial viability, a factor that fundamentally influences their interactions with the host. Despite this distinction, how bacterial viability shapes host responses remains unclear because direct comparisons using identical strains are technically challenging. Here, we systematically compared the responses induced by live and heat-treated Lactiplantibacillus plantarum 1149T using a microfluidic co-culture system with swine intestinal epithelial cells. Transcriptomics and metabolomics revealed fundamentally distinct epithelial responses depending on bacterial viability. Live L. plantarum induced a hypoxia-associated glycolytic shift and PPARG-related transcriptional responses independently of HIF1A, accompanied by the production of lipid mediators including 12,13-diHOME and 9,10-diHOME. In contrast, heat-treated L. plantarum induced nuclear factor kappa B (NFKB)-associated pro-inflammatory gene expression in a Toll-like receptor 4 gene (TLR4)-dependent manner, likely in response to alterations in bacterial surface components. Our findings demonstrate that bacterial viability influences host epithelial metabolic and immune responses, providing a mechanistic basis for the rational selection of probiotic or postbiotic strategies.

